CT colonography services look set to expand rapidly over the next five years, and considerable investment will be required to maintain performance and improve patient outcomes, according to the authors of comprehensive new practice guidelines and standards.
A group of 11 experts in the field worked together to produce a 45-page document published jointly by the British Society of Gastrointestinal Radiologists (BSGAR) and The Royal College of Radiologists (RCR). These standards can be downloaded free-of-charge from the RCR website and may be useful in supporting business cases for additional staff recruitment, say the authors, who were led by Dr. Andrew Plumb, PhD, associate professor of medical imaging at University College London (UCL) and consultant radiologist at UCL Hospitals NHS Trust.
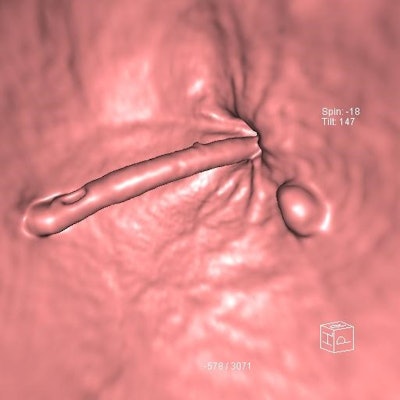
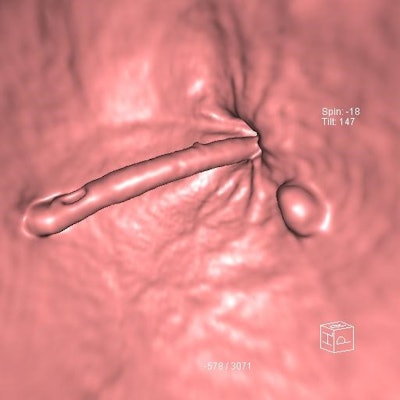
 CTC axial supine image, endoluminal image, and the corresponding colonoscopy of a polyp in the cecum. Courtesy of Dr. Andrew Plumb, PhD.
CTC axial supine image, endoluminal image, and the corresponding colonoscopy of a polyp in the cecum. Courtesy of Dr. Andrew Plumb, PhD.CT colonography (CTC) is a highly sensitive, well-tolerated test for the diagnosis of colorectal cancer and polyps, they explained. Colorectal cancer is the fourth most common cancer in the U.K., with over 42,300 cases a year. Currently, more than 100,000 CTC examinations are conducted each year in England alone, and this figure is increasing each year.
When performed to the highest quality, CTC has excellent diagnostic accuracy for clinically significant neoplasia -- i.e., colorectal cancer and advanced neoplasia -- in both symptomatic and screening populations. But unlike colonoscopy, substantial variations in practice exist in the U.K. and internationally, they added.
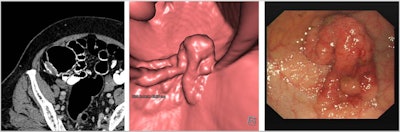
 Axial supine, axial prone, and endoluminal CTC images all show a flat cecal polyp. Courtesy of Dr. Andrew Plumb, PhD.
Axial supine, axial prone, and endoluminal CTC images all show a flat cecal polyp. Courtesy of Dr. Andrew Plumb, PhD.The guidance is divided into three parts:
- Technical and process standards ("what to do")
- Evidence-based quality measures and performance indicators for monitoring services and individuals ("what to measure")
- Practical advice, audit definitions, and templates to assist services in auditing and documenting adherence to the relevant standards ("how to measure it")
The authors noted a greater emphasis on audit and measurable metrics than in previous editions.
"Completing such audits will be time-consuming unless units adopt efficient ways to gather the data; structured reports (as are already required for the Bowel Cancer Screening Programme [BCSP] in England) will make this much easier, as the large majority of data points can be captured at the time of scan reporting," they pointed out. "Structured report templates are therefore strongly encouraged; in particular the BCSP has recommended structured reporting templates that would be suitable."
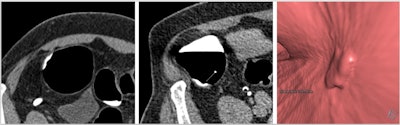
 CTC shows a 9-mm adenoma adjacent to the anorectal junction and the rectal catheter. Courtesy of Dr. Damian Tolan.
CTC shows a 9-mm adenoma adjacent to the anorectal junction and the rectal catheter. Courtesy of Dr. Damian Tolan.All standards are based on published literature where such evidence exists, and where no clear evidence is available, the guidance is derived from the opinion of the authors.
For each indicator, they identified a minimum standard. Where such standards are not met, services or individuals must take action to improve performance; where higher standards are judged desirable and have been shown to be achievable, these have been set as aspirational targets, they continued.
These targets represent the highest quality practice, and the approach mirrors the model that has been deployed to great success for colonoscopy, the authors wrote.
Ensuring patient safety
"We know there is huge variability in the way this examination is performed; this document sets out the standards which ensure delivery of the highest quality of examination and sensitivity for disease detection. This maximises patient safety while optimising the study," wrote Prof. Mark Callaway, RCR's medical director for professional practice in clinical radiology, in the foreword to the document.
The guidance uses all available evidence to define optimum examination technique and standards of reporting including both minimum numbers of examinations to be reported by an individual and the time taken to report studies. "These are the performance indicators which allow us to evaluate our own departments and individual performance," he said.
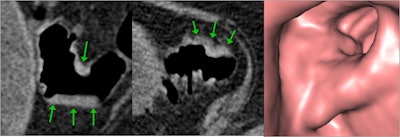 CTC demonstrates proximal sigmoid stricture with typical endoluminal features of adenocarcinoma confirmed on colonoscopy. Courtesy of Dr. Damian Tolan.
CTC demonstrates proximal sigmoid stricture with typical endoluminal features of adenocarcinoma confirmed on colonoscopy. Courtesy of Dr. Damian Tolan.He thinks the section on local audits is particularly useful because it makes it possible to turn the standards and recommendations into local service improvement. Overall, the authors have achieved a set of standards that is patient-focused and will allow the development of a consistent and high-quality CTC service, added Callaway.
The main contributors to the document were Drs. Dominic Blunt, Imperial College Healthcare NHS Trust; Ingrid Britton, University Hospital of North Staffordshire; Cindy Chew, NHS Lanarkshire; Emma Helbren, Hull University Teaching Hospitals NHS Trust; Jason Katz, patient representative; Andrew Lowe, Taunton and Somerset NHS Foundation Trust; Iain Morrison, East Kent Hospitals University NHS Foundation Trust; Andrew Plumb, PhD, (lead collaborator) and Stuart Taylor, University College London Hospitals NHS Foundation Trust; James Stephenson, University Hospitals Leicester; Damian Tolan, Leeds Teaching Hospitals NHS Trust; and Stuart Williams, Norfolk and Norwich University Hospitals.
To download a copy of the standards, go to the RCR website.










![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)





