A joint U.K. and New Zealand initiative to study cardiac defects through the use of a virtual 3D heart model offers the potential of better diagnosis and interventional treatment of cases of atrial fibrillation (AF). For the first time, researchers have identified the separate and compounding roles of electrical heterogeneity and fiber structure of the atrial tissue responsible for AF.
"Not only will studies using 3D reconstruction yield better understanding of the mechanisms behind AF, but we also hope the model can be used to test the effects of antiarrhythmia drugs that can be delivered to the heart by interventional procedures, and also to design better strategies for AF ablation," said lead investigator Henggui Zhang, PhD, a professor of biological physics at the School of Physics and Astronomy at the University of Manchester in the U.K.
![Top: Reconstructed 3D anatomy of the sheep atria with fiber structures (colored lines). Bottom: Simulated erratic electrical excitation waves during AF (cell membrane potentials are color coded from -80 mV [blue] to +30 mV [red]). All images courtesy of Henggui Zhang, PhD.](https://img.auntminnieeurope.com/files/base/smg/all/image/2013/01/ame.2013_01_29_12_03_23_100_2013_01_30_virtual_heart_Geom_Fib_top.png?auto=format%2Ccompress&fit=max&q=70&w=400) Top: Reconstructed 3D anatomy of the sheep atria with fiber structures (colored lines). Bottom: Simulated erratic electrical excitation waves during AF (cell membrane potentials are color coded from -80 mV [blue] to +30 mV [red]). All images courtesy of Henggui Zhang, PhD.
Top: Reconstructed 3D anatomy of the sheep atria with fiber structures (colored lines). Bottom: Simulated erratic electrical excitation waves during AF (cell membrane potentials are color coded from -80 mV [blue] to +30 mV [red]). All images courtesy of Henggui Zhang, PhD.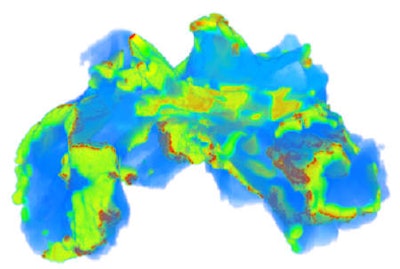
Atrial fibrillation is a common heart condition that causes an irregular heartbeat and increases the risk of stroke, with an incidence of around 1% to 1.5% in the world's population. It occurs when abnormal electrical impulses, commonly triggered in the pulmonary vein, override the heart's natural rhythm, which desynchronizes muscle contraction and lowers the heart's efficiency. With aging, this incidence rises to a range of 5% to 10%, and is expected to further increase sharply in the next two decades, according to Zhang.
"A lack of understanding as to why exactly abnormal electrical impulses occur means that until now treatment for AF has remained unsatisfactory. With greater understanding of the roles of various contributory factors, there can be far more tailored and effective management for patients," he said.
The 3D software model was created by imaging a thinly sliced sheep's heart using modalities such as MRI, diffusion tensor-MRI, and confocal microscopy. The researchers also developed a method of using micro-CT for scanning the heart with very good spatial resolution of up to about 10 µm.
 With greater understanding of the roles of various contributory factors, there can be far more tailored and effective management for AF patients, according to Henggui Zhang, PhD.
With greater understanding of the roles of various contributory factors, there can be far more tailored and effective management for AF patients, according to Henggui Zhang, PhD.
"We now have a software reconstruction of the whole heart in 3D in great anatomical detail. Simulation of the electrical action potential of single atrial cells was incorporated into this anatomical dataset to investigate the contribution of these cells' electrical heterogeneity to the genesis of AF," Zhang said.
Electrophysiological activity of atrial cells was simulated by mimicking ionic flows across cell membranes based on experimental data obtained by using "voltage clamps" techniques, with a set of mathematical equations used to describe the dynamics of ion channels operating across these membranes.
Simulated erratic electrical waves that passed through the pulmonary vein and the surrounding atrial tissue revealed that the intrinsic regional differences in the electrical activity of cells between the pulmonary vein and the left atrium play a major role in the genesis of atrial fibrillation.
Furthermore, the model was also able to discern the contributory role of anisotropic atrial fiber structure to the generation of atrial fibrillation, as this impacts electrical wave conduction. An abrupt change of fiber orientations at the junction between the left atrium and the pulmonary vein also seemed to help generate and develop atrial fibrillation, according to the study.
It is believed the model will be able to simulate various types of atrial fibrillation and how it would be depicted in different imaging contexts. Structural and functional abnormalities visualized will help doctors to understand the underlying electrical abnormality in the atria and this information could be used in computer packages to pin down factors causing atrial fibrillation, and even eventually be incorporated into computer-aided diagnosis software for clinical application, according to Zhang.
In terms of treatment testing, the effects of antiarrhythmia drugs can also be incorporated into the mathematical model. Simulation will allow doctors to determine how a drug altering the electrical waves traversing cells can help to terminate the "tornado-like" waves contributing to atrial fibrillation, according to Zhang.
"Diagnostic and interventional radiologists should be interested in the results of this research," he added.
While 3D reconstruction of a rabbit's heart and a dog's heart is ongoing in collaboration with the Auckland team in New Zealand, the sheep's heart model, finalized in January, is ready for basic and clinical applications. Doctors working with atrial fibrillation and who are interested in further details or involvement are invited to contact Zhang and his research team at [email protected].