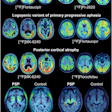
GE Healthcare announced that the first patient has been dosed in a phase III trial underway in France with a new dopamine transporter PET imaging radiopharmaceutical.
The agent is for evaluation of adult patients with suspected Parkinson's disease, and it was originally developed by French firm Zionexa, which GE's Pharmaceutical Diagnostic business acquired last year.
The company also has plans to test another dopamine transporter agent it has licensed from brain imaging firm LikeMinds in Boston for use with SPECT imaging, it said.