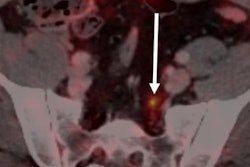
Radiopharmaceuticals firm Theragnostics is reporting encouraging results from a phase II clinical study on the efficacy of diagnostic imaging with gallium-68 (Ga-68) tris(hydroxypyridinone) prostate-specific membrane antigen (THP-PSMA) PET/CT for prostate cancer.
The prospective, open-label Ga-68 THP-PSMA PET/CT Imaging in High Risk Primary Prostate Cancer or Biochemical Recurrence of Prostate Cancer (PRONOUNCED) trial is designed to evaluate the safety and clinical impact of Ga-68 THP-PSMA PET/CT imaging on patient management and treatment decisions for men with newly diagnosed or biochemically recurrent prostate cancer after initial treatment.
Of the 50 enrolled patients in the study, treatment plans changed for one-third of the subjects with newly diagnosed prostate cancer after their respective Ga-68 THP-PSMA PET/CT scan. In addition, more than 50% of the patients with biochemically recurrent disease -- including 75% in postradical radiotherapy -- had their therapy regimen altered to a more appropriate strategy.
The results also indicated that Ga-68 THP-PSMA was well tolerated with no serious adverse events, leading the researchers to conclude the approach is safe.
"The PRONOUNCED study was completed with a substantial number of changes to the treatment regime made following the Ga-68 THP-PSMA PET/CT scan and shows how this exciting technology can influence the management of prostate cancer patients," said co-investigator Dr. Jamshed Bomanji, a professor at the Institute of Nuclear Medicine at University College London Hospitals. "Ga-68 THP-PSMA PET/CT imaging allows for prospective analysis of the clinical disease status rather than retrospective, allowing for better informed decision-making."
The trial is jointly funded by Innovate UK and Theragnostics. It is conducted at University College London Hospitals National Health Service Foundation Trust and multiple referral sites in the U.K.