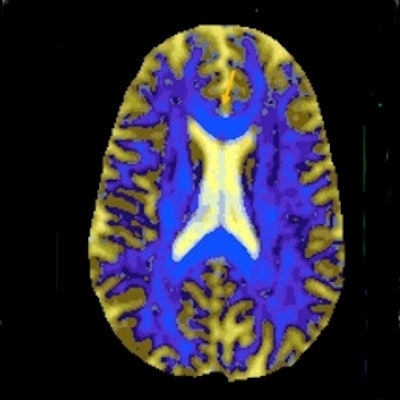
Researchers from multiple groups in the U.K., headed up at St George's University of London, have developed a single score MR imaging methodology for correlating cerebral small vessel disease (SVD) damage to cognitive decline.
The developed methodology utilizes a diffusion tensor image segmentation (DSEG) technique, combining several MRI-detectable SVD markers to produce a sensitive singular score for disease severity and brain damage. This enables monitoring of alterations in brain microstructure over time via a single angular measure -- DSEG Θ.
Diffusion tensor imaging is an MRI-based technique that has been increasingly used for neuroimaging, due to its ability to image brain tissue damage. It uses the diffusion of water molecules in the brain to generate contrast and noninvasively image volumes within the brain. This technique may enable earlier and more efficient diagnosis of SVD, with better monitoring of disease progression in affected patients. This disease alters the microvasculature of the brain, resulting in a reduced cerebral blood flow to brain tissue. The reduction in cerebral blood flow causes the cognitive decline that is commonplace in SVD.
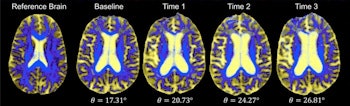
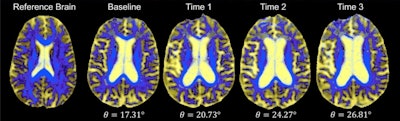
DSEG images for axial brain slices of SVD patients. Images in the SVD brain are shown over three time points with DSEG Θ angle scores for microstructure changes. Increases in Θ show substantial DSEG changes in brain microstructure over time with increasing brain damage. © 2017 The Authors (CC BY-NC-ND 4.0).
The use of DSEG markers allowed the study authors to accurately monitor changes in brain structure that correlated with cognitive testing in 98 SVD patients (ages 43 to 89) over a three-year period (NeuroImage Clinical, 15 August 2017, Vol. 16, pp. 330-342). DSEG measurements have been made previously to monitor tumor growth, but this is the first time the methodology has been used to monitor sensitive and gradual changes in brain matter.
Common imaging techniques for diagnosing and monitoring SVD only use a single marker, which does not always correlate strongly with disease progression and cognitive decline. The DSEG technique acquires scores by comparing SVD patients' brain scans with those of age-matched healthy controls. This process accurately depicts microstructure changes within the brain.
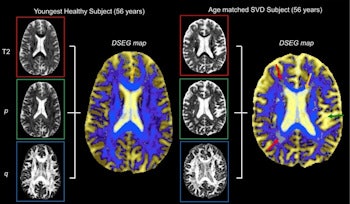
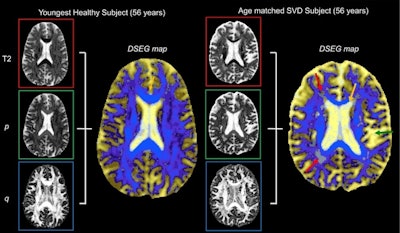
Diffusion tensor image segmentation color maps of the brain from healthy subject and age-matched SVD subject. By overlaying T2, p and q images, brain sensitive alterations in brain microstructure are visible. Red arrows indicate white matter hypersensitivities; orange arrow shows corpus callosum; green arrow shows the greater volume of cerebrospinal fluid space. © 2017 The Authors (CC BY-NC-ND 4.0).
The DSEG technique enables imaging of gray matter, white matter, cerebrospinal fluid, and white matter hypersensitivities, which when combined to create a DSEG Θ angular score can quantify brain damage effectively. The authors note a limitation of this study is the lack of consideration for the spatial location of SVD-related pathology.
Future studies could delve into the relationship between the spatial configuration of brain damage caused by SVD and the progression of cognitive decline. This imaging technique and implementation of a singular score denoting structural brain changes could be used for a range of neurodegenerative diseases -- such as vascular dementia and Alzheimer's disease -- with great promise for advancing knowledge in this research area.
Geoffrey Potjewyd is a doctoral student contributor to medicalphysicsweb, working in the division of neuroscience and experimental psychology, as part of the CDT in Regenerative Medicine at the University of Manchester in the U.K. He is studying the neurovascular unit in relation to vascular dementia and Alzheimer's disease, using biofabrication, biomaterials, and stem-cell based techniques.
© IOP Publishing Limited. Republished with permission from medicalphysicsweb, a community website covering fundamental research and emerging technologies in medical imaging and radiation therapy.










![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)





