Skipping systematic biopsy in men with elevated prostate-specific antigen (PSA) levels but negative MRI results reduces overdiagnosis by more than 50% -- and doesn't boost risk of missing clinically significant disease, researchers have reported.
The study results could help clinicians tailor care for men with elevated PSAs, wrote a team led by Dr. Jonas Hugosson, PhD, of the University of Göthenburg in Sweden. The group's findings were published on 25 September in the New England Journal of Medicine.
"Many studies have shown that men with an elevated PSA level and negative MRI results (i.e., a PI-RADS score of 1 or 2) still have a substantial risk of having clinically significant cancer detected if systematic biopsy is performed," Hugosson and colleagues wrote. "[But in] many cases, these cancers are low-volume [International Society of Urological Pathology] grade 2 cancers that are of questionable clinical significance at the time of diagnosis."
No consensus exists as to whether population-based prostate cancer screening should be recommended, according to the investigators, who noted that "the largest obstacle has been the high rate of overdiagnosis when prostate-specific antigen (PSA) is used as the identifying marker of risk." MRI for prostate cancer screening tends to have lower rates of overdiagnosis when only MRI-targeted biopsy is performed and systematic biopsy omitted, the team explained.
In an attempt to clarify MRI's role in prostate cancer screening, Hugosson and colleagues conducted an analysis of data from the GÖTEBORG-2 screening trial, the main objective of which was "to evaluate, in a long-term screening program, whether a strategy of omitting systematic biopsy in men with a PSA level of 3 ng/ml higher and performing only targeted biopsy of MRI-positive lesions would reduce the risk of detecting clinically insignificant prostate cancer while still enabling the detection of clinically significant prostate cancer at a curable stage."
The trial began in 2015 and included 38,316 men between the ages of 50 and 60 invited for PSA screening. Of these, 19,733 attended at least one screening. Men with a PSA level of 3 ng/ml or higher were recommended to have an MRI prostate exam and a systematic biopsy; those with suspicious findings on this MRI exam were recommended to have an MRI-targeted biopsy. Of the men who attended at least one screening, 6,578 were placed in a systematic biopsy group (that is, one that takes multiple tissue samples from the prostate) and 6,575 in an MRI-targeted biopsy group (performed due to suspicious lesions found on MR imaging).
The percentage of men with an indication for biopsy was 2.8% of the MRI-targeted biopsy group (elevated PSA/suspicious lesion on MRI) and 6.9% of the systematic biopsy group (all study participants with an elevated PSA level).
The authors found that the risk of detecting clinically insignificant cancer was 57% lower in the MRI-targeted biopsy group than in the systematic biopsy group (relative risk, 0.43, with 1 as reference).
| Biopsy type performance | |||
|---|---|---|---|
| Category of cancer | Systematic biopsy | MRI-targeted biopsy | Relative risk (with 1 as reference) |
| Screening-detected cancer and interval cancer, overall | |||
| Clinically insignificant | 2.4% | 1% | 0.43 |
The study results could translate to better patient care, according to Hugosson and colleagues.
"In this trial, omitting [systematic] biopsy in patients with negative MRI results eliminated more than half of diagnoses of clinically insignificant prostate cancer, and the associated risk of having incurable cancer diagnosed at screening or as interval cancer was very low," they concluded. "These results should encourage guideline committees to update recommendations around prostate cancer diagnosis and screening."
The complete study can be found here.


![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)



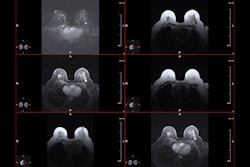
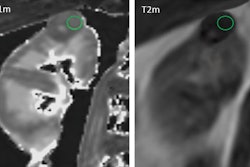


![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)