A vendor-neutral CT coronary artery calcium (CAC) score calibration tool shows considerable promise for improving heart disease risk assessment, according to a Journal of the American College of Cardiology: Cardiovascular Imaging study.
Developed by a group led by Niels van der Werf, PhD, of the University Medical Center Utrecht in the Netherlands, the score calibration tool demonstrated clinical value when tested on CT exams from the Multi-Ethnic Study on Atherosclerosis (MESA).
"MESA participants who were reclassified to a higher calcium category by means of the [calibration tool] experienced more coronary heart disease events, indicating improved risk categorization," the authors reported.
CAC scores can vary across CT systems, which can complicate heart disease risk assessment, according to the authors.
"Substantial variation in Agatston scores acquired with different computed tomography scanners may influence patient risk classification," the group noted.
In the 1990s, researchers began to use electron beam tomography (EBT) for CAC assessment. CT has now replaced this for the most part, but current CAC assessment frameworks are based on studies that used EBT. Previous studies have shown that CT CAC measurements improve risk assessment for both coronary heart disease and atherosclerotic cardiovascular disease in asymptomatic patients; it is done using the Agatston scoring method, which calculates the amount of plaque identified on CT.
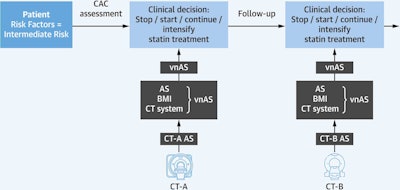 Illustration of the use of the vendor-neutral Agatston score (vnAS) calculator, which converts a multidetector CT-specific Agatston score (AS) (CT-A AS or CT-B AS) into a vnAS based on patient and CT-specific parameters. The vnAS can subsequently be used for clinical decisions on statin treatment for patients at intermediate atherosclerotic cardiovascular disease risk, both initially and in follow-up assessments. BMI 1/4 body mass index; CAC 1/4 coronary artery calcium. Courtesy of Niels van der Werf, PhD, Dr. Tim Leiner, PhD, et al and Journal of the American College of Cardiology: Cardiovascular Imaging.
Illustration of the use of the vendor-neutral Agatston score (vnAS) calculator, which converts a multidetector CT-specific Agatston score (AS) (CT-A AS or CT-B AS) into a vnAS based on patient and CT-specific parameters. The vnAS can subsequently be used for clinical decisions on statin treatment for patients at intermediate atherosclerotic cardiovascular disease risk, both initially and in follow-up assessments. BMI 1/4 body mass index; CAC 1/4 coronary artery calcium. Courtesy of Niels van der Werf, PhD, Dr. Tim Leiner, PhD, et al and Journal of the American College of Cardiology: Cardiovascular Imaging.But scoring can differ across CT systems, which is why there is a need to calibrate scores, van der Werf's group explained.
"Clinically, variations in Agatston score may translate into risk misclassification and improper treatment in up to 6.5% of asymptomatic individuals," the team wrote.
To address the problem, van der Werf's group developed a CAC score calibration tool, using two phantoms containing calcium with seven different CT and one electron-beam tomography (EBT) system (the latter of which was used for reference). The team then conducted a study that included data from 3,181 MESA participants who underwent CT exams, comparing coronary heart disease event rates between low calcium (Agatston score of less than 100) and high calcium groups (Agatston score equal to or greater than 100).
The calibration algorithm reclassified 85 low-calcium patients (11%) to a higher-risk category, and the coronary heart disease event rate in this cohort was 15% higher for these individuals compared to low-risk patients who were not reclassified (7%). The coronary heart disease hazard ratio for reclassified low calcium patients was 3.39 (reference, 1).
There's no doubt that CAC evaluation is crucial for patient care, according to van der Werf and colleagues.
"CAC evaluation is not only important for initial risk stratification but also in follow-up analysis ... [as previous research has shown] CAC progression [is] more pronounced in patients with coronary heart disease events than in those without events," they concluded.
The study was published on June 14 and can be found here.











![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)




