Using late gadolinium-enhanced MRI scans, German researchers found myocardial fibrosis, or scarring of the heart, among competitive male triathletes. An increased risk of scarring was associated with the amount of exercise performed, according to the group, who will present the study at next week's RSNA meeting in Chicago.
Although the study included both men and women, the condition was not found among the female triathletes.
"The clinical relevance of these scars is currently unclear," noted lead author Dr. Jitka Starekova, from the department of diagnostic and interventional radiology and nuclear medicine at University Medical Center Hamburg-Eppendorf, in a statement from the RSNA. "However, they might be a foundation for future heart failure and arrhythmia."
Starekova and colleagues studied a group of 55 male (average age, 44 years) and 30 female (average age, 43 years) triathletes, who underwent late gadolinium-enhanced cardiac MRI scans. The contrast agent quickly washes out of normal heart tissue but is retained for longer in scarred tissue.
Evidence of myocardial fibrosis was apparent in the left ventricle in 10 (18%) of the 55 men; the condition was not present in any (0%) of the 30 women. The athletes with scarring had completed significantly longer total, swimming, and cycling distances, and they had higher peak exercise systolic blood pressure than the subjects without myocardial fibrosis.
When the researchers examined the lifetime competition history of the athletes, they found that the number of completed Iron Man events and middle-distance triathlons was significantly higher for the men than the women, leading them to suggest that the myocardial fibrosis risk was associated with the amount of exercise.
"Comparison of the exercise test results revealed that female triathletes had lower systolic blood pressure at peak exercise and achieved lower maximal power compared to male triathletes," Starekova said. "Furthermore, comparison of the sport history showed that females had a tendency to complete shorter distances compared to male triathletes. This supports the concept that blood pressure and race distances could have an impact on formation of myocardial fibrosis."
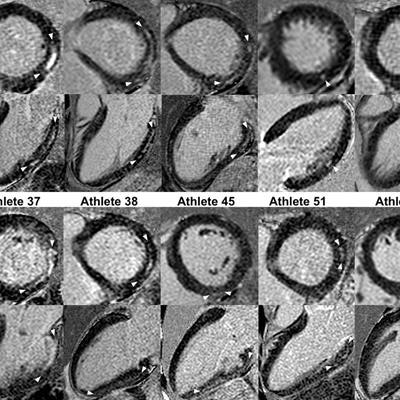
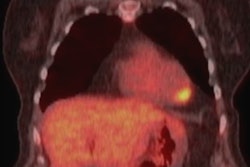
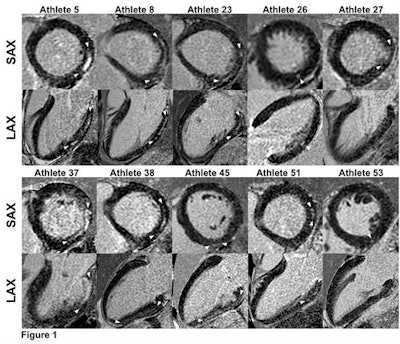 Short- and long-axis images with late gadolinium enhancement depicting a nonischemic fibrosis pattern in all 10 triathletes. The size of late gadolinium enhancement, indicated by arrows, varied from small, point-shaped (triathlete 53) to larger and more distinct (triathletes 5, 8, and 37) areas of fibrosis. Image courtesy of RSNA.
Short- and long-axis images with late gadolinium enhancement depicting a nonischemic fibrosis pattern in all 10 triathletes. The size of late gadolinium enhancement, indicated by arrows, varied from small, point-shaped (triathlete 53) to larger and more distinct (triathletes 5, 8, and 37) areas of fibrosis. Image courtesy of RSNA.The researchers cited several factors for the possible link between the amount of exercise and the risk of myocardial fibrosis. Higher exercise-induced systolic blood pressure could result in greater myocardial mass, and more exercise might expose athletes to a higher risk of myocarditis, or inflammation of the heart muscle. These factors, along with repeated increased stress of the left ventricular wall due to exercise, could injure the heart muscle.
"Although we cannot prove the exact mechanism for the development of myocardial fibrosis in triathletes, increased systolic blood pressure during exercise, the amount and extent of race distances, and unnoticed myocarditis could be cofactors in the genesis of the condition," she said. "In other words, repetition of any extreme athletic activity may not be beneficial for everyone."
Starekova and colleagues plan to conduct long-term follow-up studies to see if any cardiac events occur in the triathletes who had evidence of myocardial fibrosis.









![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)





