Whole-body diffusion-weighted MRI (DWI-MRI) can help assess treatment response in patients with myeloma and potentially reduce the number of painful bone marrow biopsies they need to undergo, according to a study published online in Radiology.
The U.K. study found that DWI-MRI showed the spread of myeloma, a type of blood cancer, throughout the bone marrow better than conventional tests.
Using DWI-MR images, experienced readers were able to identify treatment response with high sensitivity, and apparent diffusion coefficient (ADC) values were "highly repeatable and also provided good sensitivity for assessing response," the authors wrote (Radiology, January 28, 2014).
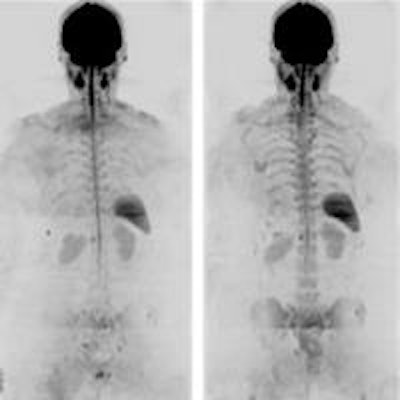
 Whole-body DWI of a 72-year-old woman with myeloma who did not respond to treatment. Images are before (left) and after (right) three cycles of treatment. Images courtesy of Radiology.
Whole-body DWI of a 72-year-old woman with myeloma who did not respond to treatment. Images are before (left) and after (right) three cycles of treatment. Images courtesy of Radiology.
The prospective study, led by Sharon Giles from Royal Marsden Hospital, included 34 myeloma patients who underwent DWI prior to treatment, with 26 of those patients undergoing post-treatment DWI-MRI. Imaging was performed on a 1.5-tesa scanner (Magnetom Avanto, Siemens Healthcare) with patients in the supine position.
Of the 26 subjects who were imaged after treatment, 21 were identified as responders: two had a complete response, five had a very good partial response, and 14 had a partial response. The other five patients were classified as nonresponders, with three remaining stable and two showing disease progression.
Based on image scores from the two experienced radiologists, sensitivity for detecting treatment response was 86% (18 of 21 patients) and specificity was 80% (4 of 5 patients).
In addition, changes in ADC correctly identified treatment response for 24 (96%) of 25 myeloma patients. A 3.3% increase helped to identify treatment response with 90% sensitivity and 100% specificity, while an 8% increase achieved 70% sensitivity and 100% specificity.
Giles and colleagues believe the study is the first time myeloma information could be gathered from all the bones in the entire body in one scan, without having to rely on individual bone x-rays. DWI-MRI allowed them to assess the degree of myeloma in individual bones and monitor treatment response.
One limitation, however, is that the skull remained difficult to image, due in part to metal dental implants and fillings.












![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)




