All the data is there: the muscle, the fat, the bone density. Every abdominal CT contains a silent readout of a patient's physiological reserve that, study after study now shows, adds prognostic information beyond what body mass index (BMI) alone can offer, including for surgical complications, chemotherapy toxicity, and mortality.
And yet, as musculoskeletal radiologist Kerensa Beekman, MD, PhD, of Radboud University Medical Center asked an audience that mostly raised no hands, how many of you are currently reporting body composition metrics in your multidisciplinary team (MDT) meetings? No one.
 "We can see what the clinicians cannot see, and especially sarcopenia and osteoporosis are under-reported and under-diagnosed," said Kerensa Beekman, MD, PhD, of Radboud University Medical Center.All pictures courtesy of Claudia Tschabuschnig
"We can see what the clinicians cannot see, and especially sarcopenia and osteoporosis are under-reported and under-diagnosed," said Kerensa Beekman, MD, PhD, of Radboud University Medical Center.All pictures courtesy of Claudia Tschabuschnig
CT is the gold standard for opportunistic body composition assessment in routine practice, stated Christian Krestan, MD, of the Medical University of Vienna. At the L3 vertebral level, a single axial slice is highly representative of whole-body muscle and fat distribution, a correlation confirmed in studies dating back to 2004, and machine learning segmentation now performs comparably to manual analysis.
The invisible predictor
The parameters that matter are skeletal muscle area (SMA), skeletal muscle index (SMI, corrected for height squared), visceral adipose tissue (VAT), subcutaneous adipose tissue (SAT), and muscle density as a proxy for fat infiltration within the muscle itself, what oncologists call myosteatosis. High VAT is strongly associated with adverse metabolic profiles, mortality in intensive care unit (ICU) patients, and worse outcomes across multiple cancer types.
Low muscle mass, or sarcopenia, is an independent predictor of postoperative complications, longer hospital stays, reduced chemotherapy tolerance, and mortality. Low bone density, readable as Hounsfield units at L1, has been shown in some cohorts to predict vertebral fracture risk at least as well as, and in certain populations better than, dual-energy x-ray absorptiometry (DEXA).
Beekman noted that nearly 30% of patients in CT-based assessments may meet sarcopenia criteria, yet the condition is still underdiagnosed and underreported. While obesity, visible on the outside, gets coded, sarcopenia, invisible until you look at the muscle, almost never does.
Beyond the tumor: What cancer does to the whole patient
In a parallel session on the holistic evaluation of cancer patients, Mario Ghosn, MD, of Hôpital Henri Mondor in Créteil, France, made the case that tumor response assessment alone tells an incomplete story. Focusing on pancreatic cancer, he showed that patients who lose 10% or more of skeletal muscle mass during chemotherapy have significantly higher mortality, regardless of how the tumor itself responds on imaging.
 "We need to standardize and automate our measurement methods," said Mario Ghosn, MD, of of Hôpital Henri Mondor in Créteil, France.All pictures courtesy of Claudia Tschabuschnig
"We need to standardize and automate our measurement methods," said Mario Ghosn, MD, of of Hôpital Henri Mondor in Créteil, France.All pictures courtesy of Claudia Tschabuschnig
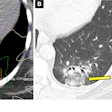
He presented a case of a patient with carcinoma of the head of the pancreas. The mass had decreased in size, enough to suggest imaging stability, and standard response assessment would have stopped there. Body composition analysis told a different story: 25% loss of muscle mass, 41% loss of visceral fat, 33% loss of subcutaneous fat.
Body composition suggested physiological deterioration despite apparently reassuring tumor imaging. This is the second readout body composition offers in oncology, not just pre-treatment risk stratification, but longitudinal monitoring of the patient alongside the tumor.
Common language still missing
Standardization remains the field's central limitation. Different studies use different slice levels, different Hounsfield unit thresholds, and different cutoff values for sarcopenia. Male and female populations differ significantly, as do Asian and Western populations. Ghosn named the consequence: without agreed methods, the field cannot reliably compare results, apply cutoffs across centers, or build the evidence base needed for clinical adoption.
The surgeon needs to know
Beekman's contribution on the clinical integration question was practical and a little uncomfortable. She is actively trying to raise body composition at MDTs. The reception has been mixed.
High visceral fat is associated with up to twice the risk of post-operative complications, and with increased likelihood of a laparotomy instead of a laparoscopic approach, a clinically significant difference in recovery trajectory. And yet BMI, which cannot distinguish muscle from fat or tell you anything about visceral fat distribution, remains the tool most clinicians reach for.
Her proposal is incremental but specific. Report fatty infiltration in muscle when you see it, even informally, mild, moderate, severe. Measure L1 Hounsfield units when bone looks thin, report vertebral fractures. It takes seconds and is on every scan.
Fix the fracture, but look at the muscle
Danoob Dalili, MD, of Oxford University Hospitals, presented on vertebroplasty and fragility fractures, a tighter technical lens. Vertebral compression fractures affect close to one million people annually in the U.S. alone and are likely more prevalent and expensive in Europe and Asia.
 "Drug dependency is a growing problem and also can lead to sarcopenia, especially when patients are put into bed very early," said Danoob Dalili, MD, of Oxford University Hospitals in Oxford, U.K.All pictures courtesy of Claudia Tschabuschnig
"Drug dependency is a growing problem and also can lead to sarcopenia, especially when patients are put into bed very early," said Danoob Dalili, MD, of Oxford University Hospitals in Oxford, U.K.All pictures courtesy of Claudia Tschabuschnig
Large observational cohorts have reported very high long-term mortality after vertebral compression fracture, around 43% by four years in Medicare-based analyses, and as high as 85% at 10 years in later claims-based data. A meta-analysis published in Radiology of more than two million patients found that those treated with vertebral augmentation were 22% less likely to die than patients managed conservatively over a 10-year follow-up period.
The body composition connection is clinically intuitive rather than fully settled: immobilization after fragility fracture can accelerate muscle loss and frailty, especially in already sarcopenic patients, and prolonged bed rest may compound the very vulnerability that contributed to the fracture in the first place.
Treatment toxicity: An underappreciated link
Stefania Rizzo, MD, of the Oncology Institute of Southern Switzerland in Lugano, presented on what may be the least visible body composition effect in oncology: its relationship to treatment-related toxicity.
Chemotherapy doses are currently calculated by body surface area, a formula based only on height and weight that takes no account of how much of that weight is muscle, how much is fat, or how it is distributed.
In endometrial and cervical cancer, sarcopenia and myosteatosis at diagnosis predict toxicity, treatment delays, dose reductions, and early discontinuation. In one study of endometrial cancer patients receiving carboplatin and paclitaxel, lean body mass measured by CT was a better predictor of paclitaxel-induced peripheral neuropathy than body surface area.
The evidence is accumulating across cancer types, but the field still lacks standardized thresholds, automated integration into routine reporting workflows, and a reimbursement framework that makes the extra effort viable at scale. These barriers are partly structural, around workflow, automation, and reimbursement, and partly unresolved methodological ones, particularly around standardization of cutoffs and scan parameters across populations and centers.
Reading what's already there
Ghosn put the challenge in one frame: a radiologist producing a beautiful, detailed lung cancer CT report, every lesion measured, every node described, with no mention whatsoever of the muscle wasting visible in the same scan. AI segmentation now matches manual analysis, and fully automated solutions are emerging that can extract visceral and subcutaneous fat, skeletal muscle area, aortic calcium, and bone density from a single routine CT scan. The technical tools are no longer the obstacle, but the workflow is.
If roughly 30% of oncology patients have sarcopenia that goes undiagnosed and unreported, that is not a gap in the science. It is a gap in the workflow. The next standard of care for cancer patients may not be a new imaging technique, but reading what's already there.