4DMedical has received CE Mark certification for its CT:VQ imaging software, enabling commercial launch across the EU.
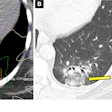
CT:VQ combines the anatomical detail of CT with functional ventilation-perfusion mapping from routine non-contrast CT scans, providing quantitative regional lung function insights without the need for radiotracers or nuclear medicine infrastructure, according to the company. The software is designed to fit within existing CT-based workflows, the firm said.
In a concurrent announcement, 4DMedical noted that it has secured $83 million Australian dollars (€49.7 million) in a private placement from institutional investors to support European expansion and other growth initiatives.
CT:VQ has received FDA clearance and is currently deployed at six U.S. academic medical centers, including Stanford, Cleveland Clinic, and Mayo Clinic, according to 4DMedical. The company said it is now advancing commercial engagement plans across Europe.