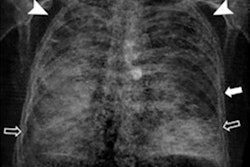
Dark-field chest radiography may be superior to conventional chest x-rays for diagnosing early emphysema, according to preliminary findings from an early clinical trial that were presented at ECR 2022 Overture.
German researchers compared the performance of a prototype dark-field chest radiography system against conventional x-ray for diagnosing mild, moderate, and severe emphysema. Radiologist readers were better able to identify mild emphysema when reading dark-field images, the group found.
"Dark-field radiography increases the diagnostic value of attenuation-based radiography for the identification and staging of emphysema, especially in the early stages," said Theresa Urban, a doctoral candidate in biomedical physics of the Technical University of Munich (TUM).
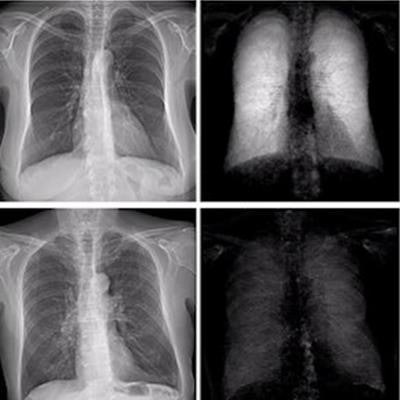
Dark-field chest radiography adds a new layer of information to conventional radiography. The technique exploits the physical wavelike properties of x-rays by directing beams through custom-made grating interferometers.
Image contrast is formed by multiple refractions of the x-ray beam at small interfaces (small-angle scattering), such as between water and air in the alveoli of the lungs. Since the interferometers are designed to be added to existing machines, the technique provides regular attenuation-based information as well as dark-field x-ray contrast images in single acquisitions.
The technique recently emerged from lab studies to the clinical stage of development. In the study presented at ECR 2022 Overture, the group sought to evaluate its diagnostic performance head-to-head for the first time in a clinical trial compared to conventional x-rays.
The researchers enrolled 91 patients who underwent CT scans for suspected emphysema and who were diagnosed at varying stages, from no emphysema to mild, moderate, and advanced destructive disease. Dark-field and attenuation-based radiographs were acquired simultaneously in the patients at a radiation dose of 0.035 mSv.
For each patient, three radiologist readers then evaluated conventional x-ray images alone, dark-field images alone, or both images displayed together for the presence and severity of disease. The CT scans served as ground truth.
 Doctoral candidate Theresa Urban of the Technical University of Munich presented the first clinical trial evidence evaluating dark-field chest radiography during ECR 2022 Overture.
Doctoral candidate Theresa Urban of the Technical University of Munich presented the first clinical trial evidence evaluating dark-field chest radiography during ECR 2022 Overture.The dark-field images showed varying signal strengths based on the severity of emphysema, which confirmed its sensitivity for detecting the extent of damage in alveoli microstructures.
There were no significant differences among readers in differentiating between mild to moderate and moderate to severe cases of emphysema on either conventional images or dark-field imaging, she said.
However, results of a receiver operating characteristic (ROC) analysis demonstrated that readers were better able to identify trace versus mild emphysema when reading dark-field images compared with conventional images, with areas under the curve (AUC) of 0.86 versus 0.73, Urban said.
"When the readers only had dark-field images available, or dark-field and attenuation images, there was a differentiation possible between trace and mild emphysema, which was not possible using only attenuation images," she said.
Limits of the study included the small number of patients, Urban said, adding during a Q&A session that the group at TUM also has shown that dark-field signals are stronger in patients with larger lung volumes because there are more air-to-tissue interfaces in the x-ray beam path in these patients. Yet patient weight does not affect the path of the beam, she said.
Ultimately, the goal is not to develop dark-field x-ray as a stand-alone imaging modality, but one that complements conventional x-ray systems, Urbana said.
"We always get both conventional and dark-field images from one acquisition, so it makes sense to use both modalities for the diagnosis," she concluded.


![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)




![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)








