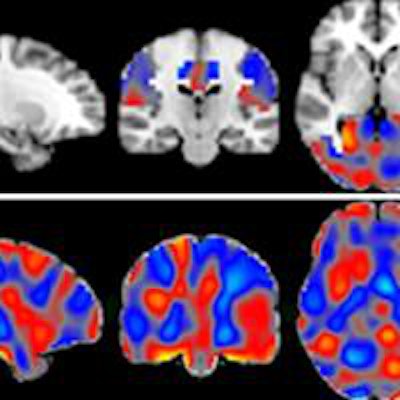
Could techniques like machine learning help radiologists diagnose Alzheimer's disease? Dutch researchers used machine-learning software to analyze arterial spin-labeled MRI scans for signs of abnormal perfusion that could be a predictor of Alzheimer's, according to a study published on July 6 in Radiology.
Alzheimer's disease is known to be a gradual process during which the brain experiences major functional changes prior to structural changes -- such as atrophy of the hippocampus -- that can be detected on imaging exams. Unfortunately, there is currently no way for clinicians to identify people with early dementia, or to tell which individuals with mild cognitive impairment will progress to Alzheimer's disease.
A research team led by Alle Meije Wink, PhD, from VU University Medical Center Amsterdam used machine-learning techniques to analyze arterial spin-labeled (ASL) MRI scans, which create perfusion maps that depict blood flow to various regions of the brain. A machine-learning application was trained to recognize patterns in the maps to distinguish between patients with varying levels of cognitive impairment and to predict the stage of Alzheimer's disease cases that were not yet diagnosed (Radiology, July 6, 2016).
Wink and colleagues studied a subgroup of 260 participants from a dementia cohort at the university's Alzheimer Center who underwent ASL-MRI scans with a 3-tesla scanner (Signa HDxt, GE Healthcare) between October 2010 and November 2012. The group included 100 patients with probable Alzheimer's disease, 60 patients with mild cognitive impairment (MCI), 100 patients with subjective cognitive decline (SCD), and 26 healthy controls.
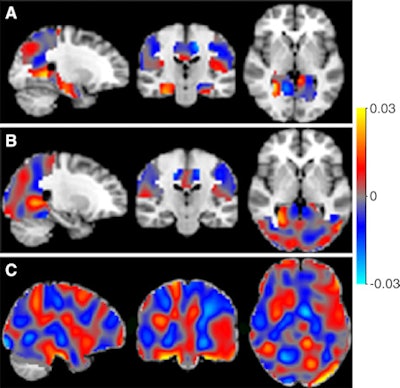 The classifiers can be represented as discrimination maps, where red indicates that the intensity at that location contributes to the likelihood of the images belonging to the more-advanced stage, and blue to the likelihood of belonging to the less-advanced stage. A: Alzheimer's disease (AD) vs. SCD; B: AD vs. MCI; and C: MCI vs. SCD. Image courtesy of RSNA.
The classifiers can be represented as discrimination maps, where red indicates that the intensity at that location contributes to the likelihood of the images belonging to the more-advanced stage, and blue to the likelihood of belonging to the less-advanced stage. A: Alzheimer's disease (AD) vs. SCD; B: AD vs. MCI; and C: MCI vs. SCD. Image courtesy of RSNA.The researchers then analyzed images using an open-access software platform, Pattern Recognition for Neuroimaging Toolbox (Pronto), which enables multivariate analysis of neuroimaging data based on machine learning.
The Pronto application was able to differentiate between individuals with Alzheimer's disease, MCI, and SCD, the researchers reported. It was also able to predict the prognosis of patients with accuracy ranging from 82% to 90%, leading Wink et al to conclude that ASL is a "promising alternative functional biomarker" for the early diagnosis of Alzheimer's disease.
What's more, automated machine-learning methods could be used as a possible screening tool by identifying brain changes on ASL-MRI scans that occur early in the disease process, they concluded.








![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)






