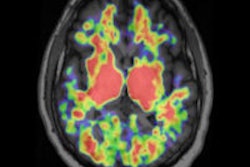
PET images show that amyloid plaque buildup in one brain region can impair brain cell metabolism and activity in another remote brain region, even though that area is not affected by amyloid plaque accumulation.
The findings, published in the June issue of Brain Connectivity, come from German researchers at Technische Universtät München in Munich and University Hospital of Cologne who used PET with FDG and the Pittsburgh Compound B (PiB) radiopharmaceutical to study parts of the same functional network that are located remotely from each other.
Lead author Dr. Elisabeth Klupp and colleagues suggest this long-distance effect may be the result of diminished neuronal signals originating from the amyloid-affected brain region to the remote amyloid-unaffected brain region (Brain Connectivity, Vol. 4:5, pp. 371-383).
In patients with Alzheimer's disease, hypometabolism has been observed in brain regions with minor amyloid load. Previous studies have been unable to confirm that the hypometabolism-only areas are a consequence of local amyloid toxicity.
So, the aim of this study was to explore whether the hypometabolism-only phenomenon may be related to pathologies in functionally connected, remote brain regions.
Researchers enrolled 19 Alzheimer's patients and 15 matched control subjects for PiB-PET and FDG/PET. Comparisons were performed to obtain maps of significantly elevated amyloid levels and reduced cerebral glucose metabolism in patients. A hypometabolism-only (HO) area was identified by calculating a subject's hypometabolism minus the amyloid burden.
The hypometabolism-only area then was used for a functional connectivity analysis in resting-state functional MRI data of 17 elderly healthy controls. The resulting connectivity network (HO-ICN) was retransferred into the brains of Alzheimer's patients to analyze pathologies within this network in PET datasets.
The most prominent hypometabolism-only area was detected in the left middle frontal gyrus of Alzheimer's patients. The HO-ICN in healthy controls showed a major overlap with brain areas significantly affected by both amyloid deposition and hypometabolism in patients.
Researchers concluded that the results support the notion that hypometabolism in brain regions not strongly affected by amyloid pathology may be related to ongoing pathologies in remote but functionally connected regions.










![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)




