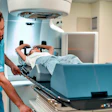
Radiation therapy firm Varian Medical Systems has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for two of the company's products.
The FDA cleared the latest version of its Vitesse real-time planning system for high-dose-rate (HDR) brachytherapy. The agency also cleared Varian's Edge radiosurgery suite, a system for advanced radiosurgery using real-time tumor tracking and motion management technologies. In addition to receiving FDA 510(k) clearance for Vitesse, Varian also obtained the CE Mark.
With the latest version of Vitesse, HDR brachytherapy treatment plans can be created in real-time, using ultrasound images generated in the operating room rather than CT scans generated elsewhere.
Varian also has taken an order for three medical linear accelerators from the Algerian Ministry of Health.
The Varian Clinac iX devices, along with Eclipse treatment planning software and Aria oncology information management software, will be located at a newly constructed facility to deliver treatments such as intensity-modulated radiotherapy and image-guided radiotherapy.