When participants at a training course run by the European Society of Breast Imaging (EUSOBI) were presented with a case study and asked to provide their first-line assessment using an Apple iPad, the instant poll revealed the challenge of changing clinical practice for assessing whether a patient complaint of nipple discharge is symptomatic of cancer.
Approximately 41% of the audience called for ductography, while 33% responded that a cytological analysis of discharge fluid would be their first choice. Fewer than 20% of participants in the course considered an MRI examination, a new and emergent response presented by Dr. Anna Linda, from the Institute of Diagnostic Radiology at the University of Udine in Italy.
Nipple discharge is the third most common breast complaint seen by physicians, and in 8% to 15% of cases, it is associated with a breast cancer, she reported. While ductography is effective in locating and identifying intraductal lesions, she said its main limitation is that it is unable to distinguish between benign and malignant pathology for the abnormality. It is an invasive procedure as well, fraught with potential complications.
"The limited ability of ductography led to a search for a more effective, noninvasive imaging method to assess this condition," said Linda. "MRI was found to offer an alternative diagnostic modality in patients with negative results from conventional imaging."
 "New emergent indications: B3 at needle biopsy and nipple discharge" was the theme of Dr. Anna Linda's presentation at the EUSOBI course.
"New emergent indications: B3 at needle biopsy and nipple discharge" was the theme of Dr. Anna Linda's presentation at the EUSOBI course.
MRI using a protocol that combines T2-weighted images without contrast enhancement and then contrast-enhanced T1-weighted images results in high sensitivity and an excellent negative predictive value (NPV), according to Linda, citing results from eight studies. A retrospective review of prospectively collected cases from 37 patients at the University of Udine clinic found four malignancies and five papillomas, returning a sensitivity of 100% and an NPV of 100%.
Another area with emerging evidence for MRI as an alternative method of assessment is high-risk lesions diagnosed at core-needle biopsy.
"The question is whether MRI can determine preoperatively the presence or absence of concurrent malignancy with B3 lesions and potentially guide a decision for clinical radiological follow-up, as opposed to surgical excision," she said.
Linda is the lead author of a paper published last month in the American Journal of Roentgenology exploring whether malignancy can be ruled out safely with breast MRI (February 2012, Vol. 198:2, pp. 272-280). Across 166 patients, a total of 169 high-risk lesions were identified. After analysis of the MRI examinations, 116 lesions (68.6%) were considered negative for malignancy, and the remaining 53 lesions were considered malignant. Twenty-two lesions at surgical excision were found by histologic analysis to be malignant.
While MRI underestimated malignancy for lobular neoplasia and atypical ductal hyperplasia, the negative predictive values of MRI were very high for papilloma and radial scar. This result led the authors to conclude that patients with papilloma and radial scar and without suspicious MRI findings can safely undergo clinical and radiological follow-up instead of surgery.
Retrospectively, if this policy had been applied, 79 unnecessary surgical procedures would have been avoided at her center, Linda reported.
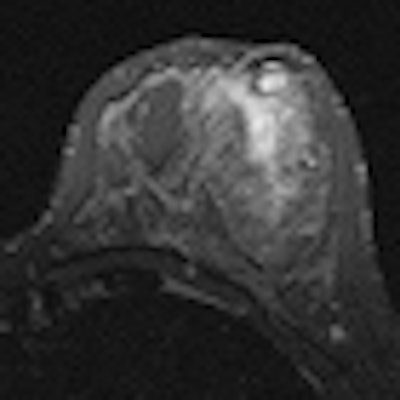
Editor's note: On the home page is a short-tau inversion recovery (STIR) T2-weighted axial breast MR image that shows suspicious nonmasslike enhancement in the inferior outer quadrant of the right breast. The final pathological diagnosis was papillary carcinoma with extensive intraductal carcinoma. Image courtesy of Dr. Michele Lorenzon, University of Udine.











![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)





