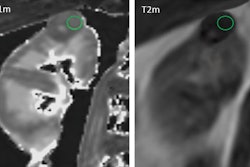
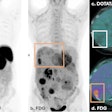
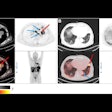
Telix Pharmaceuticals is highlighting that the first patient has been dosed with the company's TLX250-CDx (Zr-89 DFO-girentuximab) for clear cell renal cell carcinoma, the most common and aggressive form of kidney cancer.
The patient program is being conducted at Radboud University Medical Centre in Nijmegen, the Netherlands, and follows the completion of Telix's phase III ZIRCON study (Zirconium in Renal Cancer Oncology) which reported positive results, according to the firm.
Telix is moving forward toward completing a Biologics License Application submission for TLX250-CDx with the U.S. Food and Drug Administration, it said.