
Trained radiographers have achieved high accuracy in screening MRI scans to exclude brain metastases, and this has led to a change in the patient pathway at the world-famous Christie Hospital in Manchester, U.K.
"If a scan is deemed to be negative, radiographers discharge patients as normal, only contacting a radiologist for a positive scan or if they are not sure," explained Alison Kilburn, a clinical specialist radiographer at the Christie's MR Unit. "The result is an improvement to both radiographer and radiologist workflows and enhanced patient experience."
 The Christie is a 188-bed (including critical care) cancer facility serving a population of 3.2 million people across Greater Manchester and Cheshire, with 26% of patients being referred from elsewhere in the U.K. Courtesy of Mark Waugh/Alamy Stock Photo.
The Christie is a 188-bed (including critical care) cancer facility serving a population of 3.2 million people across Greater Manchester and Cheshire, with 26% of patients being referred from elsewhere in the U.K. Courtesy of Mark Waugh/Alamy Stock Photo.In an e-poster presentation made recently at the virtual UK Imaging & Oncology (UKIO) Congress, Kilburn and her colleagues provided details about the introduction of radiographer screening for brain metastases in patients with a known primary cancer.
Brain metastases are a common neurologic complication seen in 20%-40% of all patients diagnosed with cancer, they noted. In 2018, 1,643 brain MRI scans for suspected new metastatic disease were performed at the Christie. This amounted to 10.1% of all MR examinations, and a large percentage of these were done for screening purposes.
"Previously, all imaging required a review by a radiologist prior to the patient leaving the department. At our facility, the radiologists have always worked remotely from the clinical department, meaning that they are not always readily available when contacted," the authors stated.
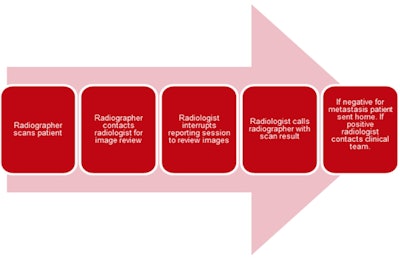 Original patient pathway at Christie Hospital. Courtesy of Alison Kilburn and UKIO 2021.
Original patient pathway at Christie Hospital. Courtesy of Alison Kilburn and UKIO 2021.The group undertook an eight-month clinical audit that included all referrals for scans to exclude or confirm new brain metastases. Following attendance at focused teaching sessions delivered by a consultant radiologist, radiographers were prospectively asked to record whether the scan was normal, metastases were present, or whether any confounding pathology was present.
A total of 429 patients were included in the audit. All were either symptomatic or asymptomatic and scanned to determine the presence of new intracranial metastatic disease. Opinions by radiographers were compared with the "gold standard" of the radiologist's final report.
For "normal" versus "abnormal," radiographers demonstrated a sensitivity of 81%, specificity of 94%, and negative predictive value -- the percentage of "normal" (negative) results deemed by radiographers that are true "normal" -- of 95%, giving "substantial agreement."
For "metastases" versus "no metastases," radiographers had a sensitivity of 98% and a specificity of 76% with negative predictive value -- the percentage of "not mets" (negative) results deemed by radiographers that are true "negative" -- of 96%, again indicating "substantial agreement."
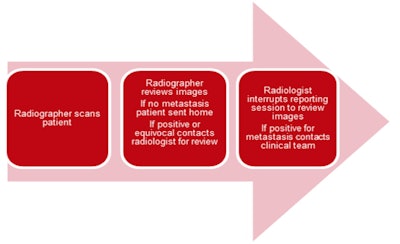 New patient pathway at Christie Hospital.
New patient pathway at Christie Hospital.Governance arrangements are in place documenting that ongoing audit and continuing education are essential in ensuring that this extended scope of practice is sustainable, transparent, and responsive to the changing workforce, the Christie team said.
"The audit has also yielded a substantial image dataset which will enhance education opportunities by allowing training specifically targeted to the areas radiographers may find problematic, such as the imaging appearances of very early disease in asymptomatic patients on surveillance," they concluded.











![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)





