Adding a liquid biopsy to MRI may improve the ability to gauge breast cancer treatment response, according to Italian research presented on 10 April at the American Association for Cancer Research (AACR) annual meeting.
Measuring plasma cell-free DNA (cfDNA) in patients after chemotherapy and prior to surgery has the potential to improve the accuracy of MRI alone in patients treated for localized breast cancer, according to the study, which was reported as a late-breaking presentation and featured in the press program at the virtual meeting.
The results suggest a possible role for liquid biopsy to bolster MRI, which has "suboptimal accuracy" for this clinical use, though further and prospective evaluation is needed, reported Dr. Francesco Ravera, PhD, a fellow in the department of internal medicine at the University of Genoa, and colleagues.
Liquid biopsy's potential for improving the sensitivity of imaging has been an important area of cancer research in recent years.
The retrospective study was sponsored by the University of Genoa and other organizations in Italy. To address the limitations of MRI as a presurgical guiding tool, researchers sought to assess if cfDNA testing could help assess pathological complete response following chemotherapy.
Typically today, sentinel lymph node biopsy is performed to assess axillary node response after chemotherapy and is followed by lymph node dissection in those who do not have a complete response. A more accurate noninvasive method for gauging complete response and for monitoring could save some patients from the need for sentinel lymph node biopsy, the researchers noted.
Researchers analyzed blood from breast cancer patients who had undergone neoadjuvant anthracycline/taxane chemotherapy, with samples available from the time of diagnosis and after treatment. They profiled the samples and based on histopathological analysis, developed a DNA integrity index as a measure of the degree of fragmentation of cells in the blood associated with responders and nonresponders.
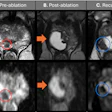
The cfDNA integrity index was evaluated in 38 patients, of whom 11 had a pathological complete response after neoadjuvant chemotherapy treatment. Researchers assessed MRI and the cfDNA integrity index individually, as well as the accuracy of the two tests together when the results were in agreement.
| Performance for predicting complete pathological response after chemotherapy | |||
| MRI | cfDNA integrity index | MRI and cfDNA (when in agreement) | |
| Accuracy | 77.1% | 81.6% | 92.6% |
Agreement of MRI and cfDNA results yielded a positive predictive value of 87.5% and a negative predictive value of 94.7%.
Ravera commented by email that researchers are currently planning a two-phase study to build on their findings. The first phase will evaluate the value of the cfDNA integrity measure and MRI for assessing complete response in a similar population of breast cancer patients, but with a larger sample size. If that phase is successful, researchers will follow up with a study of the clinical utility of MRI/cfDNA integrity index for assessing complete response, replacing sentinel lymph node biopsy with axillary ultrasound to evaluate the local relapse rate, he said.