
Ultrasound was used to track deep vein thrombosis (DVT) prevalence in patients treated with mechanical ventilation for COVID-19 in a new study. Consistent sonography helped the team identify the benefit of twice-daily prophylactic medication, according to a research letter published on 20 November.
A team at Lariboisière Hospital in Paris found that patients treated with increased anticoagulation therapy had a significantly lower prevalence of proximal DVT compared with those who received standard treatment. In a Journal of Infection letter to the editor, the team said a major strength of the study was the use of systemic ultrasound screening in all patients.
"To our knowledge, this is the first study comparing [standard prophylactic anticoagulation] with [increased anticoagulation] strategies and showing a significant reduction in femoral/popliteal DVT using systemic ultrasound screening," wrote the authors, led by Dr. Sebastian Voicu from the department of medical and toxicological resuscitation.
The new, exploratory study compared DVT prevalence before and after the hospital's intensive care unit (ICU) increased anticoagulation therapy regimens for patients with COVID-19. Approximately 10% of all COVID-19 deaths are caused by pulmonary embolism complicating DVT, according to the authors.
The first group of 50 patients underwent COVID-19 treatment before 2 April and received standard prophylactic therapy of 40 mg of enoxaparin or unfractionated heparin once per day. Meanwhile, the second group of 43 patients underwent treatment on or after 2 April and received increased prophylactic therapy of 40 mg of enoxaparin twice per day or therapeutic treatment with 1 mg/kg twice daily enoxaparin or unfractionated heparin.
All 93 patients in the study received a systematic ultrasound scan two days after undergoing tracheal intubation. A certified sonographer performed a second ultrasound scan for patients without signs of DVT on the initial scan.
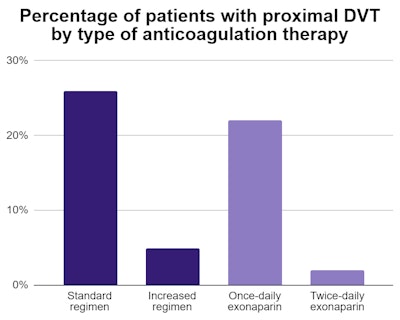
Patients in the increased anticoagulation regimen group experienced significantly fewer occurrences of femoral or popliteal DVT. Only two patients in the increased treatment group had DVT on ultrasound scans, compared with 13 patients in the standard treatment group.
The results were also significant for 67 patients specifically treated with 40 mg of enoxaparin. Only one patient treated with twice-daily enoxaparin had femoral or popliteal DVT on ultrasound scans, compared with 11 patients who received once-daily treatment.
It's important to note that the group treated with increased anticoagulation regimens was seen later in the year and, therefore, was more likely to receive dexamethasone and azithromycin. However, the authors did adjust for factors that differed between groups, and even then, anticoagulant treatment still showed a significant association with DVT prevalence.
In addition, while the study highlighted the benefit of systemic ultrasound scans for monitoring DVT prevalence, the modality couldn't answer all of the team's questions. Notably, DVT occurrence below the popliteal level did not differ between the groups, and there were not enough patients for the team to assess the effect of anticoagulation treatment on mortality outcomes.
"The favorable risk/benefit ratio of prophylactic double-dose enoxaparin 40 mg twice-daily regimen is worth exploring in future studies," the authors concluded.







![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)






