Researchers in Italy are saving time in their use of cardiac MRI to assess myocardial viability by using native T1-weighted mapping instead of a cumbersome T2-weighted imaging approach, according to a new study that compared both methods and found the latter wanting.
In 24 patients who underwent MRI within a week of suffering an ST-elevated myocardial infarction (STEMI), the team from University of Rome La Sapienza scanned patients with protocols that included modified look-locker inversion-recovery (MOLLI) imaging, short-tau inversion recovery (STIR), T2-weighted imaging, and cine MR.
They measured six regions of interest in the T1 images in order to compare T1 with T2. The researchers examined acquisition times and MR values for the standard T2 protocol including localizer, STIR, cine MR, and late gadolinium enhancement (LGE) with that of the native T1 mapping protocol that included MOLLI and cine MR.
The results showed significant differences in T1 native values between necrotic areas of the myocardium, areas at risk, and the remote myocardium. Clear differences were also seen in the comparison of T1 values between all regions (p < 0.01 for all). Finally, acquisition took about half the time for T1 versus the T2 approach.
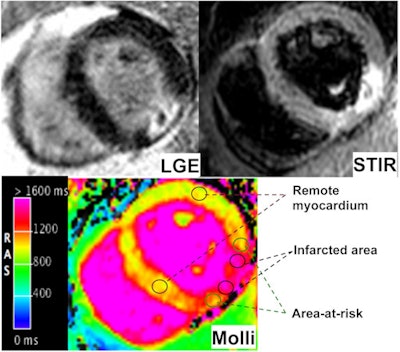 Patient with myocardial infarction seen with late gadolinium enhancement (upper left), STIR, upper right, and T1 MOLLI image depicting infracted area, area at risk, and remote myocardium. All images courtesy of Dr. Nicola Galea.
Patient with myocardial infarction seen with late gadolinium enhancement (upper left), STIR, upper right, and T1 MOLLI image depicting infracted area, area at risk, and remote myocardium. All images courtesy of Dr. Nicola Galea."There is a significant difference between the three groups with no overlap between the area of infarction and the other two areas ... and we can say that the T1 protocol is shorter than the traditional protocol," Dr. Nicola Galea said in a presentation at RSNA 2015.
Cardiac MR (CMR) is the reference standard for assessment of myocardial damage after acute myocardial infarction, he noted. The conventional approach requires a combined evaluation of myocardial edema and necrosis using T2-weighted MRI and late gadolinium enhancement, but the standard technique is time-consuming and not well tolerated by all patients.
"In particular, the late gadolinium enhancement involves a contrast injection that should be avoided in patents with chronic rental failure or with a history of repeated gadolinium injection," Galea said. "Moreover, the image analysis has a nonobjective threshold for mapping and can be affected by technical factors... Image quality depends on patient cooperation."
On the other hand, T1 mapping is an objective and reproducible method and there is no need for contrast injection, and it can further characterize the extent of ischemic injury and quantify the severity of damage, as well as predict long-term functional recovery, he continued.
A previous paper hinted at the discriminative power of T1-weighted imaging, which showed higher values than corresponding T2 images in areas of infarct, Galea said. "Furthermore, T1 values increased with the early severity of ischemic damage."
This study sought to explore the phenomenon further by examining the capability of native T1 mapping to differentiate three key myocardial regions: infarcted area, healthy myocardium, and areas-at-risk, and compare them with the conventional T2 CMR sequence as a reference standard.
The group recruited 24 consecutive patients (mean age 54 ± nine years, 68% male) with CMR within seven days after STEMI. The 1.5-tesla CMR protocol included MOLLI, STIR, T2-weighted imaging, and cine MR sequences, and inversion recovery-prepared turbo spin-echo (IR-TSE) T1-weighted images were acquired for late gadolinium enhancement after administration of the contrast agent gadobenate dimeglumine (Gd-BOPTA, Bracco Diagnostics).
The team used dedicated software (Cvi42, Circle) to analyze MOLLI images, placing four regions of interest within necrotic areas. Results were expressed using mean ± standard deviation and compared with the Student's t test.
"For image analysis, we started with a traditional method based on threshold and in particular, we applied the threshold through standard deviation for late gadolinium enhancement images," Galea said. "After delineating the infarct area we identified the area at risk by the hyperintensity of T2 images. After generation of the T1 maps, we calculated six regions of interest and in myocardium calculated the infarct area excluding the area of microvascular obstruction."
The average time to reperfusion was around four and a half hours after symptoms onset, he said. The average infarct size was around 24% of the myocardium, and the area at risk around 29%, he added.
According to Galea, "Calculating the study time of acquisition using traditional protocol for only the mapping sequence plus the cine MR sequence, we can say the T1 protocol minimum is shorter than the traditional protocol," -- 22.4 ± 8.7 minutes for T1 versus 45.4 ± 9.5 minutes for T2 (p < 0.01).
The mean T1 native value for all patients was 1,317 ± 66 msec in the necrotic area (LGE + MVO-), 1,149 ± 57 msec in the area-at-risk (LGE-/STIR+), and 952 ± 76 msec in the remote myocardium (LGE-/STIR-). There were significant differences in the comparison of T1 values between all regions (p < 0.0 l). Infarct size was 25 ± 12% of the left ventricular mass.
In the T1 results, "there is a significant difference between the three groups with no overlap between the area of infarction and the other two areas, there's a little overlap between the area at risk and remote myocardium, but no significant difference," he said. "The correlation analysis showed an increase of T1 in the infarct zone that was directly correlated with infarct size after reperfusion."
Limitations of the study included the small sample size, and T1 values in the peripheral zone were not considered.
"In conclusion, T1 mapping enables a quantitative assessment of myocardial damage compared with traditional T2-weigthted LGE sequence," Galea said. "The T1 weighting increased with the severity of the T1 damage, and provides a complete assessment of myocardial viability for the reduction of study duration without the use of a contrast agent."
An audience member said the results were promising but asked how the results could be expanded to determine the infarct area based on T1 quantification. Galea said conclusive results would require more cases.
"Our goal is to obtain stratification of the myocardium based on T1 value, but we need more patients to identify the specificity and sensitivity of those values and a specific cutoff," he explained. "At the moment, the cohort is too small with just 24 patients, but this is our goal."