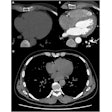
For brain tumor patients undergoing surgery, the proportion of tumor removed is a major factor determining prognosis. However, diseased tissue may look like healthy brain, making it difficult for surgeons to locate the tumor margin. Meanwhile, histological analysis of excised tissue can take 10s of minutes, extending the patient's time in theater. Expensive in-theater MRI has also shown limited benefit.
Addressing this, researchers in the U.S. developed optical imaging technology for the rapid, objective assessment of tissue samples during surgery (see Raman imaging spots brain cancer). Stimulated Raman scattering (SRS) microscopy images tissue architecture on a cellular level, and an automated machine learning classifier scores the images for the likelihood of tumor infiltration. In their latest study, the collaboration used this approach to accurately classify biopsy samples from 22 neurosurgery patients (Science Translational Medicine, 14 October 2015, Vol. 7:309, pp. 309ra163).
"This technology has the potential to eliminate some of the guesswork of brain tumor surgery," said Daniel Orringer, senior author and neurosurgeon at the University of Michigan in Ann Arbor. In identifying tumor margins more accurately, SRS microscopy could spare healthy brain from unnecessary excision and minimize or delay tumor recurrence and treatment failure, Orringer added.
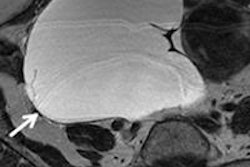
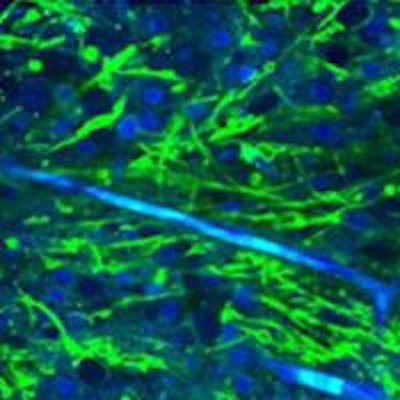
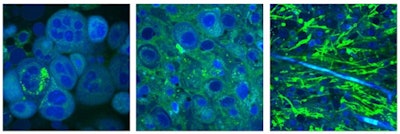 Stimulated Raman scattering (SRS) microscopy images. Left: brain metastasis from a lung carcinoma; center: human glioblastoma cells; right: infiltrative low grade oligodendroglioma, with tumor cells and an associated "chicken wire" blood vessel seen in blue. Credit: Orringer and Xie Labs.
Stimulated Raman scattering (SRS) microscopy images. Left: brain metastasis from a lung carcinoma; center: human glioblastoma cells; right: infiltrative low grade oligodendroglioma, with tumor cells and an associated "chicken wire" blood vessel seen in blue. Credit: Orringer and Xie Labs.Aiming for the release of a commercial device within the next 18-24 months, the technology is being developed by a multipartner collaboration. Partners include Harvard University, where SRS microscopy was developed by the Xie group, and start-up company Invenio Imaging. The approach stimulates Raman scattering using two lasers whose difference in frequency is tuned to match the vibrational modes of lipids and proteins. The resulting images display lipids in green and proteins in blue, enabling protein-rich tumor cells to be detected.
The tissue classifier combines three indicators of tumor infiltration extracted from the SRS images -- tissue cellularity, axon density, and a protein-to-lipid ratio -- into a single score. Ranging from zero to one, a higher score indicates a higher probability of tumor infiltration. The classifier generates the score using a generalized additive model. The model is trained using an SRS image database of normal and diseased tissue independently assessed for tumor infiltration using conventional hematoxylin and eosin (H&E) microscopy.
The researchers used SRS microscopy to image samples from 19 patients with glioblastoma and three with epilepsy. As expected, there were marked qualitative differences between the two groups and between different types of tumor and normal brain tissue. Overall, tumor-infiltrated tissue exhibited higher cellularity, a protein-rich composition, and a lower density of axons. In a blinded online survey of three neuropathologists, the researchers demonstrated the utility of the images in detecting and identifying the degree of tumor infiltration matched that of H&E staining. They obtained very good agreement between the two techniques in a sample of seven patients.
A subset of 1,477 images from 18 patients was used to assess the classifier. The classifier was trained with one-half of the images and used to score the other half. It performed well, identifying tumor-infiltrated tissue with an average sensitivity of 97.5% and specificity of 98.5%.
By combining imaging with a quantitative score, the technique has an advantage over quantitative approaches such as Raman spectroscopy, Orringer said. "As a surgeon, I'm much more likely to trust a numerical readout that makes sense given the tissue architecture. SRS microscopy allows surgeons a metric they can trust and verify."
In ongoing work, the collaboration is developing SRS microscopes suitable for clinical use, including a handheld probe for in situ assessment of tissue during surgery. Though the probe may be more convenient than ex vivo analysis, testing is needed to assess its performance in the more demanding measurement environment of the surgical cavity.
"We are working on understanding the trade-offs in image quality that might come with the handheld imaging system," Orringer said. "Once we have both an ex vivo and in vivo system available to test side by side, we'll determine which system -- or both -- we'd like to put our energy into developing."
© IOP Publishing Limited. Republished with permission from medicalphysicsweb, a community website covering fundamental research and emerging technologies in medical imaging and radiation therapy.