In a test of imaging efficiency, German researchers using a digital radiography (DR) system with a wireless detector cut 25% off the exam time for a typical single-view skeletal exam compared with conventional computed radiography.
Patients will probably not notice a difference, but it adds up to 10 extra days of scanning time available annually on the flat-table bucky system, which is what the comparison was made with at the Johann Wolfgang Goethe-University Hospital in Frankfurt am Main, Germany.
The findings were strong enough to influence equipment purchasing plans at the teaching hospital, according principal investigator Dr. Thomas Lehnert.
"We are starting out in four or five months from now with a mobile DR system. It will replace a CR system in our intensive care unit," he said in an interview with AuntMinnieEurope.com. "In the future, we will replace CR with DR totally."
Lehnert, a senior physician of radiology, elaborated on results of a study he presented in March at the 2011 ECR. His group's time-and-motion analysis of a portable cassette-sized wireless flat-panel DR detector (DRX-1 Carestream Health) was compared with conventional CR (Fujifilm Medical Systems), while both systems operated separately on a table-based radiography system (Diagnost Optimus Bucky, Philips Healthcare).
Data from 474 CR skeletal exams were collected; the CR cassette was then traded out for the wireless flat-panel DRX-1 detector to acquire an identical mix of 467 skeletal x-ray procedures. More than 50 patients were examined for each of nine types of inpatient and outpatient skeletal studies performed from January to June 2010.
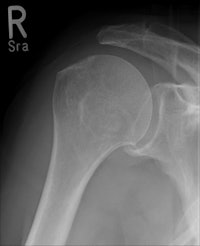
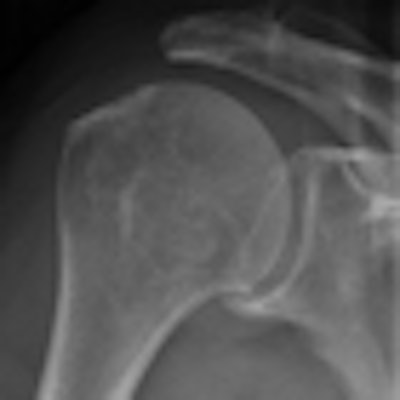
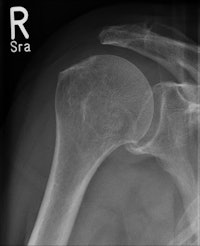 Wireless digital radiography (DRX-1) is faster and produces higher quality bone studies than conventional radiography, according to a study presented at the 2011 ECR. Conventional radiography image is at left, while wireless DRX-1 image is at right. Images courtesy of Dr. Thomas Lehnert.
Wireless digital radiography (DRX-1) is faster and produces higher quality bone studies than conventional radiography, according to a study presented at the 2011 ECR. Conventional radiography image is at left, while wireless DRX-1 image is at right. Images courtesy of Dr. Thomas Lehnert.CR cassettes were hand-carried to a reader for processing and transferred to a PACS for radiologist interpretation. DR image data were transmitted via wireless connection to an independent workstation for annotation and postprocessing before transfer to the PACS.
Total exam time for a single-view study averaged 78.93 seconds for wireless DRX-1 compared with about 105.37 seconds for CR (p < 0.001), an improvement of 24.5%, Lehnert said.
Most of the 26.44-second difference was attributed to faster postacquisition processing with DRX-1. About 13 seconds were saved by eliminating the need for cassette transport. Another 10 seconds were saved during cassette readout, and 3.2 seconds were shaved off the typical initial image processing time.
Overall, the wireless DRX-1 approach cut 38.5 minutes per day from exam times. Extrapolated results suggest that a conversion from CR to wireless DRX-1 will save about 10 days of scanning time annually, Lehnert said.
No operational problems with DRX-1 were reported. Higher equipment acquisition costs and the 30 seconds of downtime for changing an electric battery in the cassette after every 80 studies were the only negatives identified for the wireless DR approach, he said.
Lehnert and colleagues also found that image quality also significantly improved in the shift from skeletal CR to DRX-1. Those results will be described in a separate study.

















![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)