Dirtier than a bus pole or a public toilet? Ultrasound probes have come under the spotlight again in European best practice recommendations on infection prevention and control.
The guidance from the European Society of Radiology Ultrasound Working Group seeks to address apparent confusion among ultrasound practitioners with respect to the level of decontamination necessary, and when transducer covers and sterile ultrasound gel should be used -- discrepancies that were revealed in a recent European survey.
Speaking to AuntMinnieEurope.com, lead author Dr. Christiane Nyhsen, a radiologist based in Strasbourg, France, noted the main infection prevention steps are as follows:
 Dr. Christiane Nyhsen.
Dr. Christiane Nyhsen.- Transducers must be cleaned and disinfected before first use and after every examination (as the practitioner cannot know which patients may carry transmissible pathogens).
- Cleaning ultrasound probes first is important as disinfectants will otherwise not penetrate deep enough, allowing pathogens to survive. Even sterilization will be ineffective without prior thorough cleaning.
- Subsequent low-level disinfection is sufficient for standard ultrasound on intact skin as the infection transmission risk is relatively low.
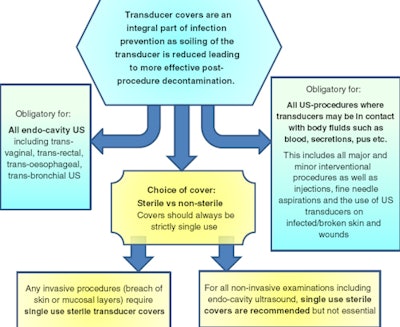
- Subsequent high level disinfection is mandatory for endocavity ultrasound and all interventions. This applies to all cases in which the transducer comes into contact with bodily fluids or when the skin is breached during procedures due to the contamination risk with bloodborne viruses, among other pathogens, which have even been isolated on acupuncture needles.
- Dedicated transducer covers must be used for endocavity ultrasound and all interventions. This reduces contamination levels significantly, but subsequent cleaning and high-level disinfection remain essential due to high reported cover perforation rates and inevitable contamination risk during cover removal.
- Ultrasound gel is not sterile unless stated on the closed sachet. Sterile gel should be used for all endocavity ultrasound and all interventions.
- Good standards of basic hygiene (in particular hand washing, protective clothing, waste disposal, etc.) and thorough decontamination of all ultrasound equipment and the environment with regular deep cleaning are mandatory.
Staff training
The authors reviewed a large number of publications including a study by Sartoretti and colleagues published in European Radiology in 2017, which found that ultrasound transducers hosted more bacteria than bus stop poles or public toilets.
"This is certainly a worrying read for all parents bringing their newborn baby for a harmless ultrasound examination! But the same study also proved that thorough hygiene training of ultrasound staff can easily eliminate this risk, making ultrasound machines not only macroscopically pleasant but also microscopically clean," noted Nyhsen, who is a member of the editorial advisory board of AuntMinnieEurope.com.
The evidence
The actual risk is very difficult to determine, and risk analysis publications are contradictory, Nyhsen continued.
"Very few cases of actual infection transmission in the ultrasound setting have been published concerning hepatitis C infections, with the exact route of transmission being uncertain. However, I personally know of cases throughout Europe that have not been published, and there will be a number that are not symptomatic or have not been attributed to a prior ultrasound procedure. I fear there will never be published strong evidence out there, but this does not mean that cross-contamination through ultrasound is nonexistent," she stated.
Hepatitis C seems to pose a particular risk, needing only a very small inoculation size of virus particles to transmit the infection, she noted, pointing to the difficulties of cleaning the fine bore of a needle guide.
"A few particles in a reused and suboptimally cleaned -- even if sterilized -- small needle guide can be sufficient to cause cross infection, it appears. Therefore, single-use needle guides are strongly recommended," she said.
Persistent bugs
HPV has been shown to be partly or completely resistant to alcohol, so using alcohol-based disinfectants to clean transducers after transvaginal procedures is not effective. Furthermore, HPV prevalence is high and the virus is closely linked to cervical cancer, as well as anogenital and oropharyngeal cancers.
Some pathogens have surprisingly long survival times, Nyhsen noted. Research from Kramer and colleagues showed that many bacteria can survive for weeks and months on dry, inert surfaces, and even fungi such as candida may still be alive after 120 days. Therefore, regular deep cleaning of the entire environment is important. Any mobile devices also should be checked and decontaminated prior to initial and after every ultrasound procedure.
| Table showing survival of pathogens on dry inanimate surfaces (shortened from Kramer et al, BMC Infectious Diseases, 2006) |
|
| Type of pathogen | Duration of persistence |
| Bacteria | |
| Campylobacter jejuni | Up to 6 days |
| Clostridium difficile (spores) | 5 months |
| Escherichia coli | 1.5 hours - 16 months |
| Haemophilus influenzae | 12 days |
| Mycobacterium tuberculosis | 1 day - 4 months |
| Neisseria gonorrhoeae | 1-3 days |
| Pseudomonas aeruginosa | 6 hours - 16 months (dry floor up to 5 weeks) |
| Staphylococcus aureus, including MRSA | 7 days - 7 months |
| Fungi | |
| Candida albicans | 1-120 days |
| Viruses | |
| Severe acute respiratory syndrome (SARS)-associated virus | 72-96 hours |
| Hepatitis A | 2 hours - 60 days |
| Hepatitis B | > 1 week |
| HIV | > 7 days |
| Herpes simplex virus 1 & 2 | 4.5 hours - 8 weeks |
| Papillomavirus | 16 > 7 days |
| Rotavirus | 6-60 days |
"It sounds great to plug an ultrasound transducer into your phone, but having a vector of Pseudomonas that survive up to 16 months in your pocket sounds somehow less appealing, doesn't it?" she said.
Legal safeguards
As workload and patient throughput increase continuously, efficiency savings are in every manager's briefing. Unsurprisingly, infection prevention is not high on the agenda, Nyhsen noted. But what if things go wrong?
"To my knowledge, there are no high-profile cases of a doctor being sued for not cleaning a standard transducer correctly -- yet. Patients who died or became seriously ill after endoscopic ultrasound procedures are a different matter; several outbreaks have been reported and hit the press," she said. "I think it is a question of time. Hygiene is increasingly in the news and patient groups are becoming more aware of the issue."
 Ultrasound staff will need to prove strict cleanliness protocols were followed if patients become ill after procedures and take their cases to court.
Ultrasound staff will need to prove strict cleanliness protocols were followed if patients become ill after procedures and take their cases to court.Inevitably complications in ultrasound may occur. However, if a case goes to court then the judge will surely inquire which infection-prevention and control protocols are in place and which standard precautions are ensured, Nyhsen stressed, pointing out that being too busy to clean a transducer will not be acceptable.
"Good staff training records and documented high hygiene standards tailored to the individual setting will be very helpful then," she noted.
Future systems
High-level disinfection with multistep wipes use effective but strong substances that may cause side effects for staff and patients, Nyhsen noted. Automated systems, which still require prior manual transducer cleaning, have the advantage of using mostly nontoxic processes. The initial investment in the machines, and the further cost of buying additional probes, will represent substantial outlay, but it is believed these systems will be significantly cheaper than multistep wipes in the long run.
"We are aware that the recommendations from our group set the bar very high and that not all practices will have the means to implement these fully at present. However, we do hope that raising awareness of infection prevention issues, encouraging everybody to do what they can within their means, and developing strict protocols will contribute significantly to raising hygiene standards," she said. "It is not only about having the best automated equipment, but understanding the importance of the different decontamination steps that will make a huge difference."
For more details, refer to the group's open access article published by Insights into Imaging in late 2017.