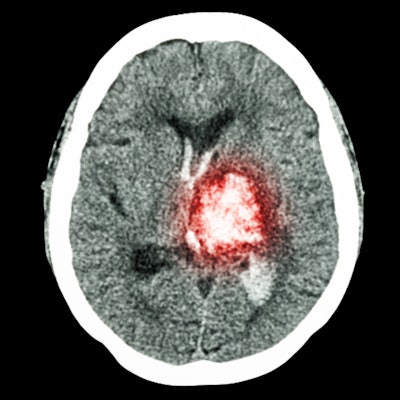
Performing angiography immediately in stroke patients improves treatment time and recovery compared with waiting for a CT scan to be conducted to guide therapy, according to research presented at the American Stroke Association's International Stroke Conference 2021.
Typically, stroke patients are imaged with CT to determine the best course of treatment, but this can result in delays. Instead, it's better to send those patients directly to the angiography suite, according to a research team led by Manuel Requena, PhD, a neurologist and neurointerventionalist fellow at Vall d'Hebron Hospital in Barcelona, Spain,
From 2018 to 2020, 150 patients who were suspected to have blocked blood vessels were randomly assigned to receive either immediate angiography or a CT scan within six hours of admission to Vall d'Hebron Hospital.
In the angiography arm, stroke patients underwent testing within 19 minutes of entering the hospital, whereas CT patients were imaged within 43 minutes. Additionally, patients in the angiography arm received endovascular treatment to restore blood flow 54 minutes faster, on average, compared with patients in the CT arm.
After 90 days, angiography patients were more likely than CT patients to demonstrate a 1-point improvement on the 6-point scale that measures stroke disability, according to Requena and colleagues. For instance, those patients didn't need assistance with daily activities.
The study was the first clinical trial examining what happens when stroke patients are transferred directly to the angiography suite, according to the researchers. Now the data must be confirmed in ongoing, multicenter, international trials.









![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)





