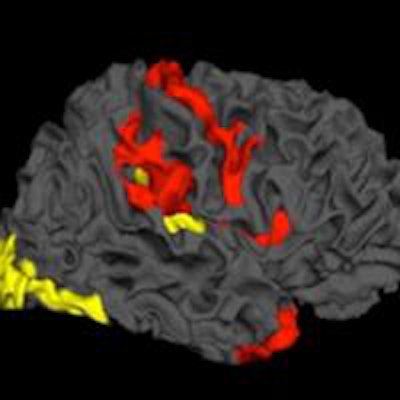
The brains of patients with schizophrenia have the capacity to reorganize themselves and fight the disease, concludes a new MRI study of brain tissue changes published online last week in Psychological Medicine.
The study included nearly 200 patients, half with and half without schizophrenia, who received MRI scans. Data from the scans were analyzed with a technique called covariance analysis to reveal tissue changes -- changes the authors said had not been demonstrated before, due to their subtlety and distributed nature.
The results showed that the brains of schizophrenic patients are continually attempting to reorganize themselves and add tissue where needed, and that improvement and reversal of the disease is possible, reported the international study team (Psychol Med, May 26, 2016).
"Our results highlight that despite the severity of tissue damage, the brain of a patient with schizophrenia is constantly attempting to reorganize itself, possibly to rescue itself or limit the damage," said Dr. Lena Palaniyappan, PhD, medical director at the Prevention and Early Intervention Program for Psychoses (PEPP) at London Health Sciences Centre (LHSC).
This conclusion runs counter to a longstanding notion that schizophrenia is a degenerative disease that only worsens over time, with therapeutic interventions aimed at slowing its progress.
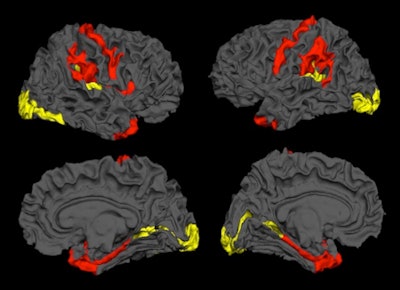 Image credit: Dr. Lena Palaniyappan, PhD.
Image credit: Dr. Lena Palaniyappan, PhD.The research comes from a collaboration among scientists from Nottingham, U.K.; Shanghai and Changsha in China; and the Robarts Research Institute at Western University and Lawson Health Research Institute in London, Ontario, Canada.
The findings are important not only because of their novelty and the study design, but also because they reveal a path forward for the development of targeted treatments that could potentially address some of the core pathology in schizophrenia, commented Dr. Jeffrey Reiss from LHSC. The development of new therapies related to brain plasticity could lead to a new sense of optimism for a disease that was traditionally described as premature dementia for its progressive deterioration, he said.
The findings "may lead us to be able to harness the brain's own compensatory changes in the face of this illness and improve recovery," said Dr. Paul Links, chair of psychiatry at LHSC.









![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)




