
Radiation oncologists are hailing the benefits of MRI-guided radiotherapy (MR-IGRT), having published a quality assurance (QA) program for their system. They claim the system is allowing them to hone their treatment margins and adapt to real-time changes in a patient's body.
"As of this month, we have treated 200 patients, with sites ranging from brain to breast to abdomen to pelvis," said Olga Green of the Washington University School of Medicine in St Louis. "We regularly observe things we've never seen before, prior to having real-time magnetic resonance imaging."
MRI-guided radiotherapy (MR-IGRT) involves the integration of 2D observations, helping a practitioner direct a radiotherapy beam to where it is needed, even when there are critical organs nearby and even when the patient is moving -- for example, by breathing.
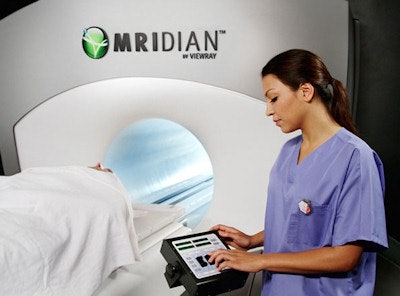 With ViewRay's MRIdian system, MRI can be used for planning and daily positioning as well as for precision targeting during treatment. Image ©ViewRay
With ViewRay's MRIdian system, MRI can be used for planning and daily positioning as well as for precision targeting during treatment. Image ©ViewRayThe first commercially available MR-IGRT system -- the cobalt-60-based system from ViewRay -- was installed at the Washington University School of Medicine and began treating patients in January 2014. Since then, three other institutions -- the University of California, Los Angeles, the University of Wisconsin in Madison, and Seoul National University Hospital -- have installed similar systems and are using them to treat patients. Only now, however, have the results of the Washington School's QA program been published (Medical Physics, 15 September 2015, Vol. 42:10, pp. 5828-5837).
As part of the QA, Green and colleagues performed four tests: a standard test by the American College of Radiology (ACR) to check image quality with use of a standardized phantom; a spatial integrity test to evaluate geometric distortion, using another phantom containing a physical grid inside; a coil test, which checks the signal-to-noise ratio of the radiofrequency coil; and a magnetic field homogeneity test, which checks the uniformity of the field inside the MRI bore using a field camera or a spherical phantom.
The results from the four tests showed that Green and colleagues' MR-IGRT system met the specifications of the ACR, the National Electrical Manufacturers Association (NEMA), and the MR-IGRT vendor. As an example, they found their measure of geometric distortion was generally minimal, except around the corners of the field. "What these results mean clinically is that when we setup our patients using MR images, we have to be mindful of these small potential spatial distortions in the corners of our images, but that the overall spatial accuracy of the system is good," Green said.
Since the system's use on patients, Green adds, they have seen the benefits of being able to reduce treatment margins, adapt to new cases daily, and track anatomy in real time. "Not only are we able to adapt to a patient's changing anatomy on the fly, we are also able to manage motion due to breathing such that we only irradiate when the target is in an optimal position," she said. "To finally be able to do this in a practical, clinical setting has been very exciting, and we have on-going clinical trials to establish the efficacy of our approaches.
"We have already treated a number of patients who would not have been able to receive radiation therapy at all with the traditional linear accelerator technology, and we are sure this new modality is a game-changer for the radiation therapy field."
© IOP Publishing Limited. Republished with permission from medicalphysicsweb, a community website covering fundamental research and emerging technologies in medical imaging and radiation therapy.









![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)








