Dutch researchers have built an algorithm that identifies and quantifies coronary artery calcium (CAC) from CT scans, paving the way for faster deployment of a test that is increasingly important in measuring the likelihood of heart disease, according to a new study in the September IIEE Transactions in Medical Imaging.
The technique automatically quantifies and identifies coronary artery calcifications in noncontrast CT scans, identifying additional candidates it is less certain of for expert review.
In 530 test images, the intraclass coefficient (ICC) between reference and automatically determined coronary artery calcium ranged from 0.98 for the left anterior descending (LAD) artery to 0.69 for the left circumflex (LCX) artery.
"We have developed a system which can either score calcium in CT fully automatically, or with a desired level of expert interaction," wrote lead author Dr. Jelmer Wolterink in an email to AuntMinnieEurope.com. "The system could be used in screening settings or large-scale studies to stratify patients with no or extremely little interaction, and also in routine clinical care to determine accurate calcium scores with slightly more interaction."
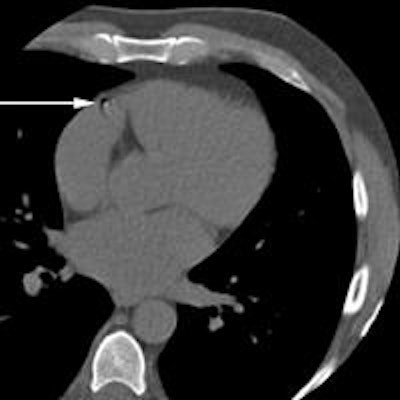
 Two examples where the system disagreed with reference annotations. Above) A motion artifact in the RCA that the system detected as a coronary calcification. Below) A coronary calcification in the distal RCA that was not identified by the system. All images courtesy of Dr. Jelmer Wolterink.
Two examples where the system disagreed with reference annotations. Above) A motion artifact in the RCA that the system detected as a coronary calcification. Below) A coronary calcification in the distal RCA that was not identified by the system. All images courtesy of Dr. Jelmer Wolterink.In addition, ambiguity detection with expert review may enable automated CAC scoring with performance comparable to that of a human expert," added Wolterink and colleagues Dr. Tim Leiner, Dr. Richard Takx, and colleagues from University Medical Center Utrecht in the Netherlands (IEEE Transactions on Medical Imaging, September 2015, Vol. 34:9, pp. 1867-1878).
Semiautomated too slow
The current seimautomated CAC test is performed using low-dose, ECG-triggered noncontarst CT. The tests aren't hard to perform, but can be time-consuming in clinical practice, screening, and large studies. This paper aimed to design an automated scoring algorithm that could do the job more efficiently.
"Automatic computer-aided identification of CAC would decrease the workload of experts," the study team wrote.
Completely automated scoring efforts aren't new. The first algorithms described candidate lesions based on location, shape, size, and intensity characteristics. But the lack of contrast between the arteries and surrounding tissues made this process challenging.
Later studies aimed to describe candidate lesions in terms of their spatial relationships to cardiac structures. A study by Išgum et al mapped probabilities for the presence of calcium, combining spatial analysis with size and intensity characteristics.
 Dr. Jelmer Wolterink from University Medical Center Utrecht.
Dr. Jelmer Wolterink from University Medical Center Utrecht.In their previous study, the Utrecht group aimed to automatically identify calcifications in routine nongated calcium scoring CT images. The system identified calcifications using a two-class classifier that sorted calcifications from negatives, or a multiclass classifier that labeled calcifications based on the associated coronary artery.
The high-intensity lesions were classified using size, shape, intensity, and location features, with locations based on a small set of coronary CT angiography (CCTA) images. Calcification candidates lacking sufficient certainty were given to experts for review, with the level of interaction determined by an ambiguity detection process based on target accuracy levels and time constraints.
This paper extends that work by detecting calcium per coronary artery, and the ambiguity detection feature has evolved to multiclass ambiguity detection, the authors wrote. It also uses noncontrast cardiac CT scans as atlases.
The authors evaluated the system using 530 test images acquired with a 256-detector-row scanner (Brilliance iCT, Philips Healthcare) acquired at 120 kVp, tube current 55 mAs, with images reconstructed to 3-mm section thickness.
For comparison, a subset of 156 scans were manually examined by a physician with five years of experience in reading calcium scans. And for 10 consecutive calcium scans that did not contain calcifications or abnormalities, corresponding CCTA scans were included in order to estimate the position of the coronary arteries, the study team wrote.
High accuracy, few ambiguous candidates
Using the two-class classifier for candidate lesions, the results showed the ICC between reference and automatically determined total coronary artery calcium volume per patient to be 0.95. Based on the multiclass classifier, the ICC between reference and automatically calculated CAC per artery was 0.98 for the left anterior descending artery (LAD), 0.69 for the left circumflex artery (LCX), and 0.95 for the right coronary artery (RCA).
The two-class classifier, which identified calcium independent of its artery, did better than the multiclass classifier, which labeled calcium based on its corresponding artery, the authors wrote.
Determining CAC burden per artery requires more classes, making it a more difficult task, the group wrote. Ranking features in the two-class and multiclass classifiers emphasize the importance of location features. The classifier takes all of those features into account to distinguish between coronary calcifications and other candidates.
"One of the main challenges is determining the exact anatomical location of potential coronary calcifications," he wrote. "It is extremely difficult if not impossible to automatically segment the coronary arteries in these images. Therefore, in this study we have estimated the position of the coronary arteries to derive spatial features, and enriched these with other features, such as texture characteristics."
There were no ambiguous candidates in 49% of all scans. Ambiguities in the remaining 51% increased the ICC for total patient coronary calcium to 1.00, and per-artery calcium to 1.00 for the LAD, 0.95 for the LCX, and 0.99 for the RCA, the team reported.
Expert review
Overall, sensitivity was 87% with an average of one 4.1-mm3 false-positive per image, and the system was able to assign patients to the proper risk category 94% of the time.
Yet, the performance of the method still fell short of a second observer, which might preclude the use of automation in clinical practice. Nevertheless, the use of ambiguity detection feature with expert review in the other 51% of scans with lower confidence showed that the system might be clinically feasible after all.
Discrimination between patients with zero and nonzero calcium scores is important, but the automated method incorrectly assigned a zero CAC score instead of a positive score or vice versa in 22 of 34 miscategorized patients, they group noted. However, the 12 patients who received a negative CAC score had low reference scores from the expert reader. The 10 patients incorrectly assigned a positive score had confounding features in the images, such as calcification in the mitral valve or noise artifacts.
"The system was able to accurately determine total patient and per artery CAC burden," the authors concluded. Still, improvements are needed.
"We believe that including more diverse training samples could help the system to better learn the differences between coronary calcifications and other candidates, and to more accurately assign coronary calcifications to specific arteries," Wolterink wrote. "We plan to perform a study with a more diverse set of patients and images to investigate how this will affect the way in which the system handles rare or ambiguous cases."










![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)





