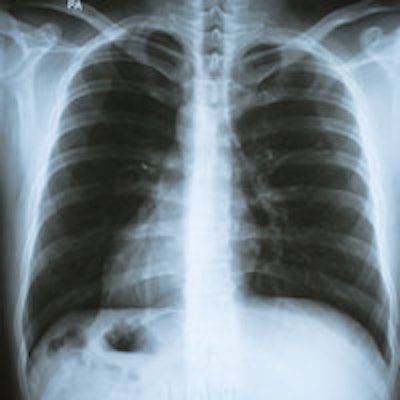
More than one-third of all lung cancer patients in the U.K. die within three months of diagnosis, despite having serial office visits before their diagnosis, according to a new report in Thorax.
The study findings suggest family doctors are missing signs of lung cancer during patient visits, and sending them home without proper investigation, the researchers wrote. What is more, the U.K. lags behind other countries in lung cancer survival, in a gap that has been largely attributed to late diagnosis of the disease, which is curable if caught in an early stage (Thorax, 13 October 2014).
The study looked at general practitioners' (GPs) investigation of lung cancer in their patients between 2000 and 2013, using data from The Health Improvement Network (THIN), which stores the anonymized health records of millions of primary care patients in the U.K.
Among 20,142 cases of lung cancer recorded by 444 general practices during the study period, only 5% or 1,071 cases were recorded on the death certificate, the study team found. Among the remaining cases, one in 10 (n = 2,036) died within a month of lung cancer diagnosis, and about one in seven (15%) died within three months. These deaths were classified as "early."
Fifty-seven percent of the lung cancer cases were in men, who were 17% more likely to die early than women with the disease, the group reported. The average age at lung cancer diagnosis was 72. Older patients, age 80 and higher, were 80% more likely to die early than those diagnosed before age 80. Current smokers were also 43% likelier to die than those who had never smoked, but interestingly, former smokers were less likely to die early than those who had never smoked, the researchers said.
Patients who died early were very likely to visit their GP more frequently in the few months before their diagnosis -- an average of five visits, the study team reported.
There were fewer cases of early death among those who had received a chest x-ray in primary care regardless of how often they had seen their primary care physician. But the chances of an early death rose in tandem with the number of doctor visits.
Also interestingly, patients registered at practices with high referral rates for chest x-rays were 41% more likely to die than practices with low x-ray referral rates, even after adjusting for other influential factors, the authors wrote, cautioning that it doesn't mean x-rays have no value, but that they may have been used at the wrong time in the wrong patient group, the study team wrote.
Finally, patients living in an area with high lung cancer rates were no more likely to survive for at least three months than patients living in an area with a low lung cancer rate, the team reported.
In an editorial accompanying the study, Dr. Michael Peake from University of Leicester's Glenfield Hospital wrote that GPs need better tools to support clinical decision-making, so they can identify more of their patients at risk for lung cancer. The excess deaths are closely related to deprivation, he said, and the gap between rich and poor keeps growing.
Peake advocated that public awareness campaigns be targeted to disadvantaged social groups to keep the gap from growing wider.










![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)




