Dutch researchers looked into automated segmentation of head and neck CT for treatment planning, and found it fast and accurate enough for daily clinical use -- dozens of times faster, in fact, than the manual technique it aims to replace.
For hyperthermia treatment planning (HTP) with CT in patients with head and neck cancers, automated segmentation of the organs in the head and neck altered the results by less than 1% compared with the time-sapping manual segmentation process currently in use. As a result, automated segmentation has completely replaced manual segmentation in practice at Erasmus University Medical Center in Rotterdam, according to the authors.
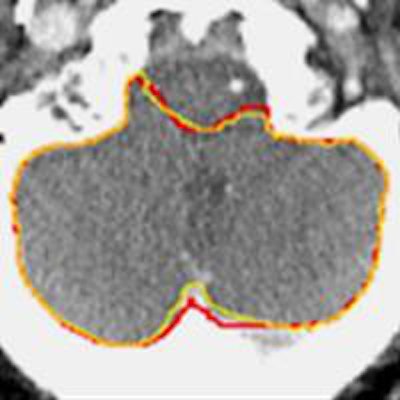
 Segmentation reveals multiple organs in the head and neck. All images courtesy of Wiro Niessen, PhD.
Segmentation reveals multiple organs in the head and neck. All images courtesy of Wiro Niessen, PhD."Compared to manual segmentation the overall change was only 1% due to interobserver variability, so we can use the algorithm to replace tedious manual segmentation," said Wiro Niessen, PhD, in a presentation at the International Symposium on Multidetector-Row CT in San Francisco in June.
Heating the tumor?
Hyperthermia treatment -- carefully heating head and neck tumors to 40° or 45° C -- has been shown to substantially improve radiotherapy outcomes in patients with head and neck cancer in several trials, even though how it works isn't fully understood, said Niessen, who is a professor of biomedical imaging at the departments of medical informatics and radiology at Erasmus University Medical Center.
"To perform this well you need to not only segment the tumor but the surrounding tumors," so as to carefully control temperature distribution during treatment, Niessen said. "To do this you need an accurate anatomic model from either MRI or CT data ... but mainly CT."
But you can't just heat first and ask questions later. Hyperthermia treatment planning in patients with head and neck tumors is essential for decision-making, treatment optimization, and treatment analysis, he said.
Previously, the only path to organ segmentation before HTP was through hours of manual segmentation work. Tissue transitions as visualized on CT have an impact on hyperthermia planning, and the problem with manual delineations is that they suffer from inter- and intraobserver variability.
Faster HTP segmentation
The Erasmus project aimed to automate the process as accurately as possible, segmenting organs of interest in the head and neck region using a fully autosegmented 3D model as an input to plan hyperthermia treatment.
Niessen and colleagues Valerio Fortunati, Jifke Veenland, and Theo van Walsum implemented a method that "combines an automatic and robust segmentation system with multiatlas registration and intensity modeling," Niessen said.
While the organs are autosegmented, the actual tumors are still delineated manually.
"We have ideas to automate this too, but it's quite complex, so what we're focused on now is delineating surrounding tissues: the eyes, the lens, the optical nerve, the thyroid -- these are the structures we would like to autosegment," he said.
The segmentation process relies on multiple atlases in which the organs have been labeled by an expert who knows the different anatomic regions, and these labeled organs can be imported. The segmentation is more robust with a larger number of atlases, so the group aims for eight or 10 to allow for wide variability in the patient's CT data. And there's another step besides segmentation.
 Atlas-based segmentation was improved by the addition of CT intensity measurements, above, in the cerebellum.
Atlas-based segmentation was improved by the addition of CT intensity measurements, above, in the cerebellum."We complemented this with intensity information," he said. "From the atlases, we also learned the appearance of different structures, and we modeled the appearance of the structures."
The spatial information from the atlases shows where to expect the structures, and the information is combined in a widely known function known as graph-cut optimization that extracts the surface from a 3D CT scan, optimizing both spatial and intensity information; this process is repeated for each region, Niessen said.
More atlases are always better to account for wider variations in anatomy. There is also technology to map one person's brain to another's, deforming and combining the results through nonrigid deformation, and inheriting the labels from the atlas, he said.
The researchers evaluated the automatically generated data versus ground truth based on manual segmentation. Testing included similarity measures, including the Dice similarity coefficient and mean surface distance. And the group tested the effectiveness of the intensity model, followed by an evaluation of accuracy and reproducibility with respect to interobserver variability. And finally, they evaluated the results with respect to hyperthermia treatment planning outcomes.
Atlas plus intensity measurements needed
"We found that atlas-based segmentation by itself is not sufficient, because sometimes there's too much anatomic variability, even with eight or 10 atlases," Niessen said. "Sometimes you need the intensity information, which does help your segmentation." The automated segmentation study was published last year in Medical Physics (July 2013, Vol. 40:7).
This year the group looked at a large dataset of about 40 cases to determine the impact of segmentation on hyperthermia treatment planning in terms of the accuracy of predicting the various anatomic structures (Radiotherapy & Oncology, April 2014, Vol. 111:1, pp. 158-163).
 Segmentation of the optical nerve and cerebellum are more accurate with the addition of intensity information (p < 0.05).
Segmentation of the optical nerve and cerebellum are more accurate with the addition of intensity information (p < 0.05).In most cases accuracy was within 1 mm to 2 mm and that was without employing intensity information, which, when added, improved results for every organ, he said.
The addition of intensity modeling had a significant impact on the accuracy of automated segmentation versus no intensity modeling (Mann-Whitney two-sided paired test, p < 0.05), according to Niessen.
Interestingly, the impact of segmentation on hyperthermia treatment owing to interobserver variability in manual segmentation was about 1% -- about equal to the 1% error encountered using automated segmentation, ultimately delivering roughly equivalent accuracy for both techniques, in addition to the time saved in every case by avoiding six to eight hours of manual segmentation, he said.
Equivalent accuracy of manual and automated segmentation in practice "means that automated segmentation can replace the time-consuming manual technique," Niessen said. "We have actually implemented it in our own cancer center."









![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)




