
Better cooperation between radiologists and rheumatologists is essential to ensure imaging is used cost-effectively to provide clinical information on diagnosis, prognosis, and serial assessment with the least risk and inconvenience to the patient, two U.K. researchers said.
"Between the specialties there are areas of mutual misunderstanding which are best resolved by a closer working relationship," noted Dr. Bernhard Tins, a radiologist at the Robert Jones and Agnes Hunt Orthopedic Hospital, Oswestry, and Dr. Robin Butler, a rheumatologist at the same hospital. "Imaging can make a huge difference for long-term patient outcome. The pros and cons of using different imaging techniques are within the remit of radiologists (otherwise what is our point?)."
Yet, it is surprising how little use some rheumatologists make of the imaging options available and how little some radiologists understand the crucial role imaging can play in determining patient outcome, they added in an article posted online on 15 October by Insights into Imaging.
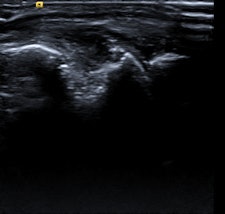
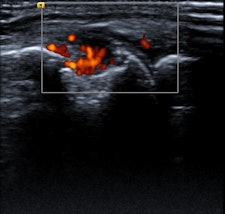 Elderly patient with established and treated rheumatoid arthritis. The patient complained of increased pain and stiffness. Ultrasound shows active synovitis and erosions in multiple metacarpo-phalangeal joints (a = grayscale image, b = power Doppler image). Ultrasound is a useful tool to assess for the presence and to an extent severity of (subclinical) inflammation. Clinical examination can be surprisingly insensitive even in florid inflammation, as shown here. Images courtesy of Dr. Bernhard Tins.
Elderly patient with established and treated rheumatoid arthritis. The patient complained of increased pain and stiffness. Ultrasound shows active synovitis and erosions in multiple metacarpo-phalangeal joints (a = grayscale image, b = power Doppler image). Ultrasound is a useful tool to assess for the presence and to an extent severity of (subclinical) inflammation. Clinical examination can be surprisingly insensitive even in florid inflammation, as shown here. Images courtesy of Dr. Bernhard Tins.Imaging in rheumatology was in the past largely confined to radiographs of the hands and sacroiliac joints helping to establish the diagnosis and then monitoring disease progression. Because radiographs are not very sensitive for early inflammation in inflammatory rheumatic disorders, the demand on imaging services was therefore limited, Tins and Butler explained. Over the last 10-15 years, new drugs and new technologies have brought fresh challenges and opportunities to rheumatology and radiology. New drug treatments allow more effective treatment, preventing many complications, and early diagnosis and disease monitoring have become the major challenges.
"The best possible patient outcome is only achieved if the two specialties understand each other's viewpoint," they wrote. "Tailored imaging examinations are best achieved by consultation of rheumatologist and radiologist ... Early diagnosis and treatment can often prevent the development of crippling disease manifestations."
In the 12-page article, the authors outline the role of imaging -- in particular radiography, MRI, CT, ultrasound, and nuclear medicine-- for the diagnosis and monitoring of rheumatological disorders, concentrating on rheumatoid arthritis, inflammatory spondylarthropathies, and gout.
Radiographs might show evidence of bone erosion and/or other reactive bone change with ankylosis of the spine, the most extreme form of reactive bone change, according to Tins and Butler. While it is important to note these findings and suggest the diagnosis if unsuspected, radiographs are of limited value in the diagnosis of early inflammatory spondyloarthropathy because of the high false-negative rate. To an even greater extent than in rheumatoid arthritis, the radiographic changes often lag behind the clinical presentation by years, they pointed out.
"Nevertheless, rheumatologists in the U.K. often request plain x-rays because the current U.K. NICE (National Institute for Health and Care Excellence) criteria for the use of TNF (tumor necrosis factor) inhibitors in ankylosing spondylitis require that the patient fulfills the modified New York radiographic criteria. While CT is more sensitive for reactive bone change, it is insensitive for edema-like change of bone marrow or soft tissues and MRI is preferred for this," they stated.









![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)





