Researchers in Germany have developed a new approach for making target treatment plans for intensity-modulated radiotherapy (IMRT) easier. They've developed a method to make this process easier, as described in the 21 March issue of Physics in Medicine and Biology.
Inverse radiotherapy planning can achieve excellent adherence to specified clinical constraints. When planning a treatment program for radiation dose delivery to tumors, it is possible to end up with "hot spots" or "cold spots," namely localized dose insufficiencies. These localized dose insufficiencies can be difficult to correct, since plan optimizations are based on global indicators such as equivalent uniform doses, for example, or dose-volume constraints for whole structures.
A "slider-based" approach offers interactive correction of hot and cold spots in IMRT treatment plans. The aim of researchers at the Fraunhofer Institute for Industrial Mathematics (ITWM) in Kaiserslautern is to use the software they developed to create a locally improved plan while retaining previously established trade-offs between critical structure sparing and target conformity (Phys Med Biol, March 2013, Vol. 58:6, pp. 1855-1867).
The current process for dealing with hot and cold spots can be arduous, said ITWM's Philipp Süss, PhD. "In a first step, the optimization parameters for the structure containing the critical spot are modified," he explained. "If the spot cannot be corrected by that, an additional volume-of-interest is defined solely for the purpose of being able to modify the dose more locally. This process is cumbersome and bears the risk of new hot or cold spots just outside the helper volume."
On-screen sliders
The approach proposed by Süss and colleagues starts by examining the initial "almost acceptable" plan generated by the treatment planning system to find any subregions with dose deficiencies. The researchers formulated a mathematical optimization problem -- the critical region correction (CRC) problem -- that obtains one or more locally corrected plans, based on the initial clinical trade-offs plus new specified dose constraints in any hot/cold spots. Unwanted hot spots are removed by applying upper bounds and local cold spots eliminated by enforcing lower bounds.
After generating the alternative plans, which are similar to the initial plan but without the local deficiencies, the planner then faces the task of deciding how much change in the initial characteristics are acceptable to obtain local improvements. Süss and colleagues suggest that this conflict should be explored interactively, and propose the use of a slider.
By manipulating on-screen sliders, users can explore trade-offs between plans while inspecting changes in the dose distribution and dose-volume histograms in real time. In cases where one alternative plan was calculated, one end of the slider position corresponds to the original plan and the other to the alternative, with positions in between corresponding to a mix of both. If more than one alternative plan is generated, additional sliders can be implemented.
Clinical case
The scheme was evaluated using a clinical example of a meningioma at the base of the skull. The original optimization problem consisted of equivalent uniform dose objectives for all risk structures, plus objectives to minimize underdose and overdose in the target.
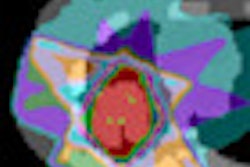
The researchers first created a plan with similar overall characteristics to that used to deliver the actual treatment. A dose slice close to the isocenter revealed a hot spot where the high dose contour entered nontarget tissue. Using prototype software developed by the ITWM team, a 9 x 9 x 9 mm subvolume with a dose of between 45.5 and 60.8 Gy was revealed. Four voxels in this region overlapped with the target contour and the remaining 23 voxels were defined as the hot spot.
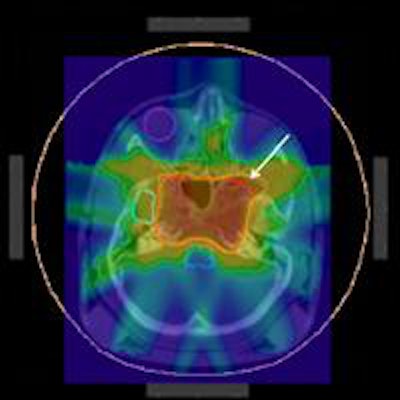
 ImageSlice of the original dose. Isolines correspond to the 50%, 90% and 95% volumes of the reference dose (set at 57.6 Gy). The marked area is the identified hot spot. All images courtesy of Philipp Süss, PhD.
ImageSlice of the original dose. Isolines correspond to the 50%, 90% and 95% volumes of the reference dose (set at 57.6 Gy). The marked area is the identified hot spot. All images courtesy of Philipp Süss, PhD.To eliminate this hot spot, the researchers solved the CRC problem, using an ambitious upper limit of 30 Gy for all 23 voxels. Two plans were generated, one using a constant voxel weighting, and the other calculated by exponentially increasing the voxel weights with increasing distance to the critical region. These plans represent extreme cases of keeping changes local and spreading the changes out.
In both cases, the critical region correction problem could be solved without violating the original underdose and overdose constraints. Dose volume histograms showed that the target curves had barely changed and that the dose in structures far from the hot spot (right optic nerve, spinal cord, and brain stem) changed significantly more in the plan where changes were allowed to spread out.
','dvPres', 'clsTopBtn', 'true' );" >
Click image to enlarge.
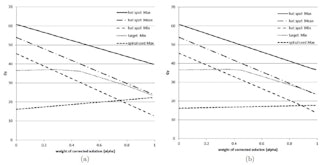
Dose values as a function of the slider values for α: (a) spread out changes; (b) keeping changes local.
The researchers then investigated the optimal mix of each two new plan and the original, using two sliders: one to control the "degree of correction" of the local insufficiencies (a slider), and another representing the "spread of allowed change" (b slider). Moving the a slider revealed the dose in the hot spot could be reduced by 10 Gy without reducing the minimum dose in the target. The maximum spinal cord dose increased significantly for plans where changes were spread over larger areas.
They also created two "compromise" plans, in which the dose was reduced by about 20 Gy. Süss noted it's also possible to create a plan that combines all three plan options, by moving both sliders. "If you image a continuous morphing between the two compromise plans, you get the right impression of the effect of pulling the b slider," he explained.
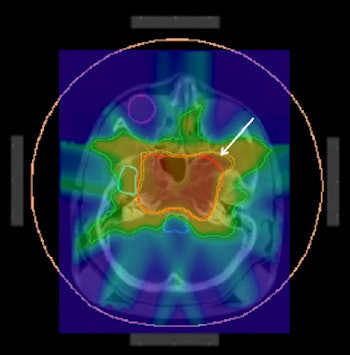
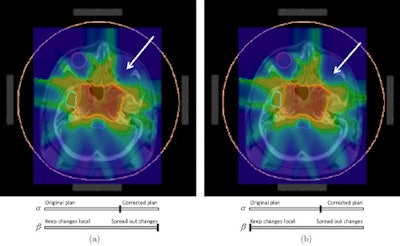 Two compromise plans: (a) spread out changes; (b) keeping changes local.
Two compromise plans: (a) spread out changes; (b) keeping changes local.The researchers conclude it is possible to eliminate a hot spot outside the target volume while controlling the dose changes to all other parts of the treatment plan. They suggest their proposed method for correcting localized dose insufficiencies could potentially become the final standard step of inverse treatment planning. "We are currently addressing how this technique can best be integrated into the overall planning process from a practitioner's view," Süss told medicalphysicsweb.
© IOP Publishing Limited. Republished with permission from medicalphysicsweb, a community website covering fundamental research and emerging technologies in medical imaging and radiation therapy.