CT can't precisely distinguish resolving from persistent and potentially malignant lung nodules at the first scan, but it does reveal the probabilities associated with different nodule characteristics, Dutch radiologists have found.
This information may also help reduce follow-up CT scans in patients known to have resolving nodules, according to the University of Groningen researchers.
Armed with data from more than 7,000 lung cancer screening subjects in the Nederlands-Leuvens Longkanker Screenings Onderzoek (NELSON) trial, the team found that a nodule's distance from the costal pleura and the margin are related to the probability that it will resolve at some point during CT follow-up. The results also showed that if a nodule is going to disappear, it will probably do so before the first three-month follow-up scan.
"Small and intermediate-sized lung nodules, say up to 10 mm, are very frequently found in CT lung cancer screening," said Dr. Rozemarijn Vliegenthart in a presentation at the RSNA 2012 meeting in Chicago. The good news is that most screen-detected nodules are benign, she said. But is there a way to differentiate benign nodules from those that need further workup? Many investigators have tried.
"We know that some of these resolve on follow-up scans," she said. "If we could predict them by CT features and distinguish and select these resolving nodules, we could potentially prevent additional CTs and workup in these patients."
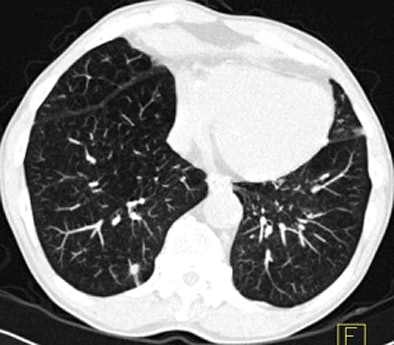 |
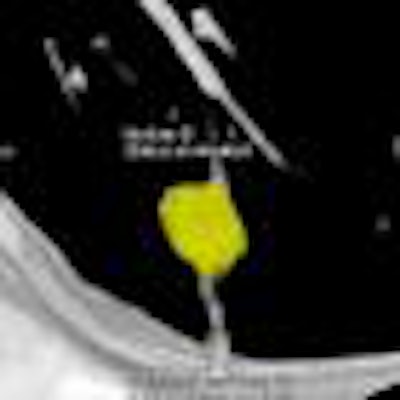
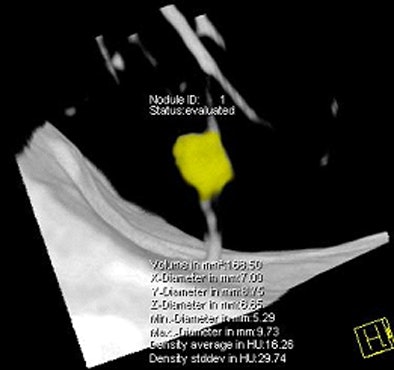
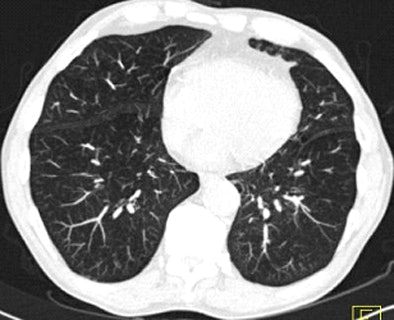
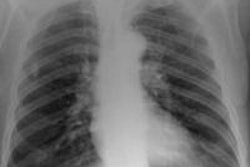
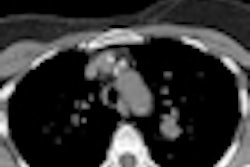
| Images above and below show a spiculated nodule measuring 169 mm3 at baseline low-dose CT screening. Bottom image, acquired four years after the baseline scan, shows that the nodule has resolved. All images courtesy of Dr. Rosemarijn Vliegenthart. |
 |
 |
Dr. Yingru Zhao, Vliegenthart, and colleagues aimed to analyze CT features associated with the disappearance of resolving nodules on low-dose CT for screening. Images for this study and the NELSON trial, which began in 2003, were acquired on one of two 16-detector-row scanners.
The analysis included solid, intraparenchymal, intermediate-sized nodules 50 mm3 to 500 mm3 (maximum, approximately 4.62 mm in diameter) visualized at baseline lung cancer screening. Indeterminate nodules were re-examined by CT at three months, and regular repeat screening CT rounds were performed after two, four, and six years, Vliegenthart said. Nodule disappearance was considered spontaneous, rather than the result of treatment. The authors compared CT features of resolving nodules with those of nonresolving, stable, and malignant nodules.
The group examined 1,059 nodules: 960 (90.7%) nodules in 730 participants did not disappear, leaving 99 nodules (9.3%) in 75 participants that resolved. In all, 75 (75.8%) resolved at three months, 17 (17.2%) resolved during the second year, five (5%) resolved at year four, and two (2%) resolved at year six.
Among the 960 nodules that remained through all follow-ups, 13 were found to be malignant, Vliegenthart said.
Nodule characteristics
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Looking at individual CT features, the group found significant differences between resolving and nonresolving nodules. Nonperipheral nodules and nodules with nonsmooth margins were more likely to resolve than peripheral and smooth nodules.
Logistic regression analysis showed no association between shape and nodule disappearance, according to the researchers.
As for margins, 56% of resolving nodules had a smooth contour, compared with 71% of the nonresolving nodules. And there were more spiculated and irregular margins in nodules that resolved, Vliegenthart said.
"Nodule distance and the margin of the nodule are related to the probability of disappearance of the lung nodule at follow-up screening," Vliegenthart concluded. "Unfortunately, these CT features overlap with [those of] malignant nodules so they cannot be used to differentiate benign from malignant nodules."
However, the finding that most nodules disappear within three months "shows the importance of short-term follow-up," she said.
The final results of the NELSON trial are due at the end of 2015.










![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnieeurope.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)





