NEW YORK (Reuters Health), Dec 12 - A reduction in breast density is a strong biomarker of response to tamoxifen chemoprevention, according to research reported Friday at the 2008 San Antonio Breast Cancer Symposium (SABCS).
"Our findings suggest that the impact of tamoxifen on risk reduction is predictable by the changes it induces in breast density after 12 to 18 months of treatment," Dr. Jack Cuzick from the Wolfson Institute of Preventive Medicine, London, and colleagues wrote in a meeting abstract.
In the previously reported International Breast Intervention Study I (IBIS-I), Cuzick and colleagues found that tamoxifen chemoprevention reduced the risk of breast cancer by up to 40% in high-risk women. They also observed that tamoxifen reduced mammographic density.
In the current analysis, they focused on whether the change in breast density after 12 to 18 months of prophylactic tamoxifen could predict the subsequent impact of tamoxifen on the development of breast cancer.
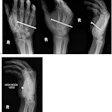
The investigators analyzed mammographic density at baseline and after 12 and 18 months of tamoxifen in 120 IBIS-I participants who developed breast cancer and 943 who did not.
Cuzick reported: "Compared to all of the women in the placebo arm, if you took the women who received tamoxifen but their breast density was reduced by less than 10%, in those women there was no benefit of tamoxifen in terms of prevention. They had the same risk of developing breast cancer as the control population."
Conversely, Cuzick said, "The women who had a 10% or more reduction in breast density -- and that was a substantial number, over 40% -- they had almost a two-thirds reduction in breast cancer" (odds ratio, 0.37).
In summary comments, Cuzick noted that "women with dense breasts typically are at four to five times the risk of developing breast cancer than women that don't have dense breasts. Mammographic density is very simple to measure and, at the moment, this information is not used very much."
By measuring density changes between baseline and follow-up mammograms in high-risk women receiving tamoxifen, "it may be possible to determine which women are actually benefiting from the intervention (and should therefore continue treatment), and those who might benefit from alternative risk-reducing strategies," Cuzick and colleagues conclude.
By Megan Rauscher
Last Updated: 2008-12-12 12:43:22 -0400 (Reuters Health)
Related Reading
Adding breast density to risk model improves cancer prediction, March 4, 2008
Copyright © 2008 Reuters Limited. All rights reserved. Republication or redistribution of Reuters content, including by framing or similar means, is expressly prohibited without the prior written consent of Reuters. Reuters shall not be liable for any errors or delays in the content, or for any actions taken in reliance thereon. Reuters and the Reuters sphere logo are registered trademarks and trademarks of the Reuters group of companies around the world.