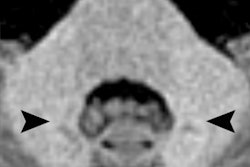
The American College of Radiology (ACR) Committee on Drugs and Contrast Media said it disagrees with the recent recommendation by a European regulatory committee to suspend the use of some linear gadolinium-based contrast agents (GBCAs) due to the potential risk of gadolinium accumulation within patients.
The ACR committee said that although intracranial gadolinium retention following intravenous GBCA administration has only recently been reported, it has been known for more than 10 years that some gadolinium chelates are not completely stable in vivo.
However, the amount of gadolinium deposited in tissues after a single GBCA dose is incredibly small and can be detected using only the most sensitive medical and scientific instruments, according to the ACR. There also remains no evidence of cellular toxicity or any credible evidence of neurologic sequelae after more than 300 million human doses worldwide.
While some studies that relied on visually observable changes in T1-weighted MRI signal have suggested that macrocyclic agents do not deposit gadolinium in brain tissues, the ACR noted that mass spectrometry data from multiple sources have confirmed that macrocyclic agents actually do deposit gadolinium within brain tissues, albeit at lower levels.
Other studies using mass spectrometry have also found varying rates of gadolinium deposition for linear and macrocyclic agents within a given class. Researchers reported that different chemical forms of gadolinium appear to be depositing within tissues, some of which would not be detectable on MRI, according to the ACR.
"Therefore, although MRI signal changes led to the observation that gadolinium was being deposited in the brain, they are less reliable for determining the quantity of gadolinium deposition in general," the ACR said in a statement. "This is particularly true for gadolinium species that are not detectable with MRI and for lower concentrations of retained gadolinium."
The ACR said it continues to endorse the need for additional research directed toward greater understanding of the mechanisms, cellular effects, and clinical consequences of gadolinium tissue deposition.
"At this time, there is no compelling evidence that any GBCAs, including linear ones, pose any safety risk with respect to brain deposition of gadolinium," the ACR wrote. "Further, linear agents have significant and well-documented diagnostic utility, and in some instances may have more desirable pharmacologic properties or a lower acute reaction risk than macrocyclic agents."
The ACR also referred interested parties to its statement with the American Society of Neuroradiology on gadolinium deposition.