Artificial intelligence (AI) chest radiography algorithms developed for use in adults currently vastly outnumber those for children, but several products are emerging in the field that appear to be promising for improving care for pediatric patients, a Dutch expert reported at ECR Overture.
Dr. Steven Schalekamp, PhD, of the Department of Medical Imaging at Radboud University Medical Center (UMC) in Nijmegen, the Netherlands, discussed the current spectrum of available AI products for chest radiography, and focused on emerging applications in pediatrics.
Currently, there are 191 AI products with the CE Mark for use in radiology, with a majority designed for neuroradiology and chest radiology (57 products from 28 vendors). Just one product, CAD4TB, which was designed to detect tuberculosis on chest x-rays, is applicable in children, Schalekamp noted in a scientific session on 2 March.
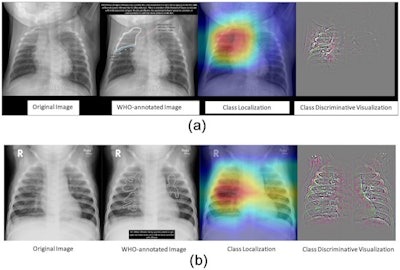 Researchers tested an AI system last year for detecting pneumonia trained on a CheXpert and PERCH dataset of 431 children. The system achieved an AUC of 0.977 for severe pneumonia. Image courtesy of PLOS One.
Researchers tested an AI system last year for detecting pneumonia trained on a CheXpert and PERCH dataset of 431 children. The system achieved an AUC of 0.977 for severe pneumonia. Image courtesy of PLOS One."It's being used in multiple often low-resource countries where they don't have radiologists available," he said. CAD4TB was trained on chest images from children ages 4 and older and visualizes abnormalities with heat maps.
Aside from this single CE-marked algorithm, other models are being developed for pediatric application that may aid in detecting pneumonia and managing patients with cystic fibrosis, with supporting published studies, Schalekamp said.
In 2020, South African researchers developed an algorithm called CAD4Kids for detecting pneumonia, which carries a high mortality rate in infants worldwide. The algorithm was trained on a dataset of chest x-rays from children under 5 years old.
The algorithm achieved reasonable sensitivity (76%) and specificity (80%), but the analysis revealed the system was still inferior to a reference observer, Schalekamp noted.
Last year, an international group led by Merck scientists also developed an AI algorithm for detecting severe pneumonia in children, but the researchers used a larger dataset, a World Health Organization set of radiographs from 431 children. The algorithm reached an impressive area under the curve (AUC) of 0.977 and was within the range of interobserver reliability of the reference observers in the study, Schalekamp said.
"These systems are also getting better and better," he noted.
Another area receiving significant attention in the past year is the development of AI algorithms to help evaluate cystic fibrosis on CT imaging in children. The disease may lend itself to AI algorithms, given there are easily recognized CT biomarkers that show progression or decline of symptoms such as bronchiectasis, peribronchial thickening, bronchial mucus, and others in patients undergoing treatment, Schalekamp said.
French researchers from the University of Bordeaux and U.S. colleagues at the University of Cincinnati in Ohio developed one such algorithm based on a dataset of images from patients with cystic fibrosis between the ages of 4 and 54.
"They were able to demonstrate that patients on treatment on follow-up CTs had lower amounts of these biomarkers, which was easily picked up by this AI system," Schalekamp said.
Challenges for AI to overcome in developing new algorithms for use in children include the simple fact that children's chests are still growing and the variability in patient positioning and inspiratory levels, Schalenkamp said.
Also, clinicians are looking for different abnormalities than in adults, such as pediatric pulmonary sequestration and congenital pulmonary airway malformations. Few large datasets exist, partially due to less imaging being performed in children due to radiation risks, Schalekamp added.
Finally, he warned that often vendors do not indicate whether their software is indicated for pediatric patients, which raises concerns over the potential for off-label use, he said.
Schalekamp concluded by offering a useful tool -- a website he and colleagues at Radboud UMC maintain for tracking all CE-marked products and their applications, listed by subspecialty and modality, as well as supporting literature and research studies evaluating new algorithms as they are published. The address is AIforRadiology.com.