German and Irish researchers have completed what they are calling the first sodium MR images of the heart at 7 tesla to deliver high-resolution images at acceptable scan times, according to just-published research in NMR in Biomedicine.
The 7-tesla acquisitions, facilitated by the use of a customized radiofrequency coil configuration and cardiac gating, achieved isotropic resolution of 6 mm, reported researchers from the Berlin Ultrahigh Field Facility (BUFF) in Berlin, along with co-authors from other institutions including Charité University Medicine in Berlin, the German Cancer Research Center in Berlin, and the University of Dublin.
"The technology enables for the first time sodium imaging of the human heart at 7-tesla with an unprecedented spatial resolution within clinical acceptable scan times," co-author Dr. Thoralf Niendorf told AuntMinnieEurope.com in an email. "This achievement provides encouragement for swift translation into clinical applications."
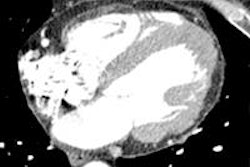
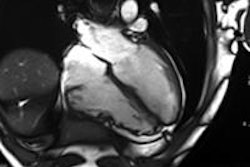
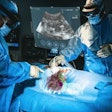
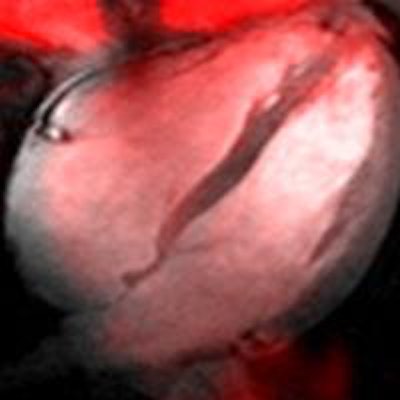
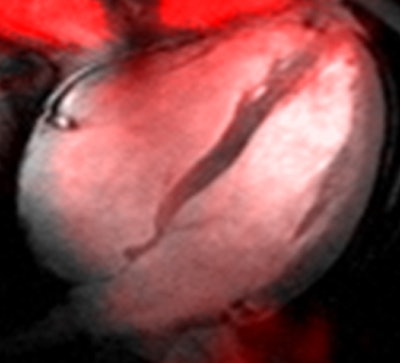 Sodium image of the human heart (in red) acquired at 7 tesla superimposed to an anatomic hydrogen MR image of a four chamber view of the heart. Image courtesy of Thoralf Niendorf, Ph.D.
Sodium image of the human heart (in red) acquired at 7 tesla superimposed to an anatomic hydrogen MR image of a four chamber view of the heart. Image courtesy of Thoralf Niendorf, Ph.D.Niendorf is a professor for experimental ultrahigh-field MR at Charité University Medicine, and heads the BUFF Max Delbrück Center for Molecular Medicine.
New view of cellular metabolism
Sodium MRI is a practical approach for improving understanding of cellular metabolism, wrote lead author Andreas Graessl and colleagues (NMR in Biomedicine, 17 June 2015).
For example, knowledge of compromised cell integrity in myocardial tissue offers valuable information for the assessment of myocardial viability, as tissue sodium concentration is a strong indicator of impaired cellular ion homeostatis and cell membrane integrity, the study team wrote.
In healthy tissue, sodium potassium adenosine triphosphatase maintains a transmembrane concentration gradient with an intracellular sodium concentration of about 15 mmol/L versus a concentration of about 140 mmol/L in the extracellular space. But during ischemia, the function of the sodium-potassium pump is degraded by declining energy reserves, leading to increased intracellular sodium concentrations, Graessl et al wrote.
Early research showed that sodium (Na-23) MRI was suitable for evaluating acute and chronic heart disease due to increased sodium concentration after myocardial infarction. However, these efforts were mostly animal studies and there were limitations imposed by the rapidly decaying Na-23 signal and extended scan times.
"Although ample signal-to-noise ratio [SNR] was demonstrated for ventricular blood, the limited myocardial Na-23 signal constitutes a challenge for cardiac Na-23 MRI en route to a clinical tool," wrote Graessl, along with Anjuli Ruehle, Helmar Waiczies, PhD, and Niendorf et al.
This study aimed to examine the feasibility of 3D and whole-heart Na-23 MRI at 7 tesla, incorporating single-cardiac-phase and cine sequences.
As their imaging protocol, the investigators used free-breathing 3D Cartesian gradient echo (GRE) and cardiac-gated 3D density-adapted radial acquisition techniques on a 7-tesla whole-body scanner (Magnetom, Siemens Healthcare).
To meet the signal acquisition requirements for the protocol, they developed a four-element transceiver radiofrequency (RF) surface coil customized for sodium MRI at 7 tesla, also simulating signal absorption rates and performing radiofrequency power deposition assessments to meet radiofrequency safety requirements.
The study aimed to demonstrate the suitability of the technique in healthy volunteers as a precursor to broader clinical studies, the team wrote.
Hardware and protocol
The dual-RF coil incorporated separate elements for sodium and hydrogen (H-1) MR signals, each containing two rectangular loop elements that share a common conductor. Each loop element was divided by chip capacitors into five elements to form a balanced and uniform current distribution along the loop, the authors wrote.
The 7-tesla scanner was equipped with a gradient system with a slew rate of 200 mT/m/ms and a maximum gradient strength of 40 mT/m. Hydrogen MR was performed with an 8-kW single-channel RF amplifier, while an 8-kW single-channel RF amplifier was used for the sodium MR signal.
Cardiac images were acquired in three healthy volunteers (two men, one woman, ages 26 to 29 years old, body mass index range 19-22). Heart rates ranged from 62 to 68 bpm. Sequences included 3D gradient echo (GRE) imaging with Cartesian phase encoding in a total acquisition time of six minutes, 16 seconds. Whole-heart coverage imaging included a density-adapted 3D radial acquisition technique that was acquired in 18 minutes, 20 seconds.
For sodium MR imaging, standard views of the heart were acquired using a nominal in-plane resolution of 0.5 x 0.5 mm2 and a slice thickness of 15 mm. For whole-heart imaging, the group achieved a nominal 6 mm isotropic spatial resolution via 3D density-adapted sodium MR acquisitions.
"This improvement versus 3D conventional GRE acquisitions reduced partial-volume effects along the slice direction and enabled retrospective image reconstruction of standard or arbitrary views of the heart," Graessi and colleagues reported.
The team applied three reconstruction modes for the 3D datasets obtained with the density-adapted 3D radial acquisition technique, including ungated reconstruction of the data, then retrospectively gated reconstruction using temporal resolution of 0.1 seconds and time increments of 0.05 seconds. This was used to reconstruct sodium MR cine images at 20 frames per cardiac cycle.
The cardiac-gated reconstructions yielded a 20% improvement in blood-myocardium contrast compared with the same data without gating, the group reported.
The coil setup showed acceptable radiofrequency characteristics, transmission field homogeneity, and sufficient depth penetration to support sodium imaging of the heart at 7 tesla acquired in clinically acceptable scan times, the authors concluded. The signal-to-noise ratio of the myocardium ranged from 10 to 27 for short-axis views of the heart.
"This achievement supports clear delineation of the myocardium, including deep lying regions of the heart," a goal that was challenging for earlier studies at 3 tesla, the authors added.
"Our in vivo studies in healthy subjects revealed that the use of a density-adapted 3D radial acquisition protocol used here provides an SNR advantage of roughly 80% compared to the used 3D GRE protocol," Graessl and colleagues wrote.
As for limitations in this feasibility study, separation of intracellular and extracellular sodium compartment is a worthy goal for future research owing to their physiological importance, the group noted.
In addition, research into suppression or selective excitation techniques would be helpful for suppressing signal contributions from blood and cartilage, which would have the additional effect of enhancing the dynamic range available for the myocardial signal, the team wrote.
Iterative reconstruction techniques that use the hydrogen imaging component to improve sodium MR component, would also be worthwhile, they added.
The path forward
The use of sodium MRI of the heart at 7 tesla is an appealing possibility for gaining insight into hypertrophic cardiomyopathy, Niendorf wrote in his email. Sodium MRI could also be useful for scrutinizing alterations in myocardial sodium concentrations caused by cardiac drugs acting on sodium ion channels, Niendorf added.
"We suggest that Na-23 cardiac MR at 7 tesla can also help to unlock questions regarding [sodium] balance and ... storage functions of myocardial tissue with the ultimate goal to provide imaging means for diagnostics and for guiding treatment decisions in cardiovascular and metabolic diseases," Niendorf wrote. "To summarize, sodium MRI of the heart at 7 tesla holds the promise to add another dimension to diagnostic imaging as a link to individualized medicine."