Top Story
Latest News
Esaote reports growth in fiscal year 2023
April 25, 2024
Smart Reporting secures €23M in financing
April 25, 2024
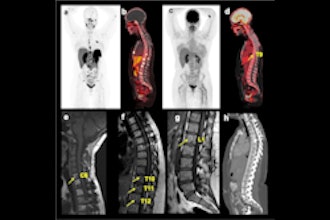
First patient dosed with ITM-31 in glioblastoma trial
April 23, 2024
Cases of the Week
Check out our Cases of the Week!
More from AuntMinnieEurope
Advanced visualization in 2024: Enhancing radiology care
April 25, 2024
ESR releases movie to mark key moments of ECR 2024
April 23, 2024
Blackford, Annalise.ai enter partnership
April 23, 2024
Anger builds over 10% fee hike for RCR members
April 23, 2024
RCR: Radiologist shortage continues
April 22, 2024
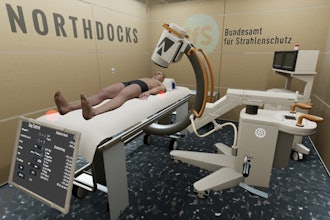
Accuray opens training facility in Switzerland
April 19, 2024
Raigmore Hospital in Scotland selects RayCare
April 17, 2024