Does virtual colonoscopy need the kind of speed that only computer-aided detection (CAD) can provide? An advanced reading technique cut interpretation times to a minimum by using CAD first for polyp detection -- followed by the radiologist, who looked only at CAD results, according to a new study from Europe and the U.S. in European Radiology.
Interpretation times were reduced to just three minutes per patient using the CAD software as a first reader, reported Dr. Thomas Mang, from Medical University of Vienna, and colleagues. The technique produced per-patient sensitivity of 100% for adenomas 6 mm and larger.
"An advanced CAD algorithm, specifically designed to be applied in an image-gallery-based first-reader workflow, can substantially decrease radiologist's reading times while enabling accurate detection of colorectal adenomas in a low-prevalence population," Mang and his colleagues wrote (Eur Radiol, 18 August 2012).
Faster interpretation times could help ensure good patient throughput as demand inevitably increases for virtual colonoscopy (also known as CT colonography or CTC) studies in the face of limited radiologist resources. One way to read more studies faster might be to shift the radiologist's efforts from "search" to "interpretation" of lesions detected using CAD as a first reader, the authors stated.
As a second reader, CAD has been shown to boost reader performance in terms of accuracy, but it comes at a cost of increased reading time, and, as a result, the use of VC CAD schemes is usually associated with slower interpretation. In a first-reader paradigm, the reader reviews only CAD marks, ignoring unprompted areas of the colonic dataset, Mang and colleagues explained.
Such a reading paradigm requires the CAD to achieve nearly 100% sensitivity on its own, because the reader has no chance of seeing lesions that the CAD missed. And it may raise medical and legal concerns stemming from the fact that the radiologist isn't seeing 100% of the colonic mucosa, only the CAD findings.
"We sought to overcome these fundamental limitations by specifically modifying the operating profile of a CAD algorithm, integrating the CAD detections into a dedicated 'first-reader' workflow based on image galleries, and complementing the overall review by adding a short 2D review of the colon to address unprompted colonic areas," the study authors explained.
Mang, Dr. Gerardo Hermosillo, Dr. Matthias Wolf, and colleagues sought to test the performance of a first-reader workflow in terms of both reading times and accuracy compared with conventional optical colonoscopy. The U.S.-based contributing institutions performed the CTC examinations with written informed consent under approval workflow for CAD detection of adenomas 6 mm and larger in a low-prevalence screening cohort.
The retrospective study looked at colonoscopy-confirmed CTC data from the landmark U.S. Department of Defense study by Pickhardt et al (New England Journal of Medicine, 4 December 2003, Vol. 349:23, pp. 2191-2120), as well as data from a study by Graser et al from the University of Munich in Germany (Gut, February 2009, Vol. 58:2, pp. 241-248). In all, 616 patients (352 men, mean age 57.8years; 264 women, mean age 57.6 years) underwent cathartic bowel cleansing and bowel insufflation prior to CT scanning.
The prototype CTC CAD system (CAD version proto_06_04, Siemens Healthcare) was designed specifically for first-reader use by increasing sensitivity at the expense of increased false-positive detections. A false-positive management approach was also employed to arrange the CAD detections in order based on system confidence of the detections, and limit the number of CAD detections for any one patient.
Based on previous evaluations of standalone CAD algorithms and published sensitivities for unassisted virtual colonoscopy reading, the authors assumed a sensitivity of 85% for adenomas 6 mm and larger. The reference standard to which CTC was compared was based on the original CTC and optical colonoscopy findings for each patient in the original study data.
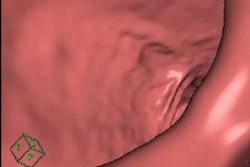
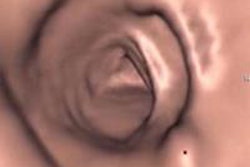
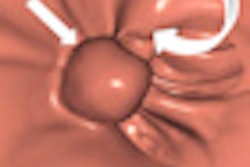
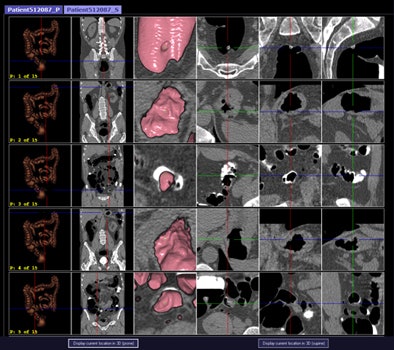 CAD first-reader gallery evaluation shown in a screening patient with a 10-mm tubular adenoma in the rectum. Above (left monitor), all 15 CAD markers in a volume are presented to the reader, line by line, as a collection of CT image galleries. They are sorted top to bottom by decreasing level of confidence, as computed by the CAD algorithm both for the prone (shown) or for the supine (not shown) position. CAD finding P:1 (first row of images) displays the polyp (crosshair) in all six gallery images. Note that the typical imaging characteristics of a polyp are already recognizable within the gallery images. The remaining CAD findings P:2-P:5 (image rows 2-5) display false-positive detections in the stomach (P:2), on the residual fluid layer (P:3), in the stomach (P:4), and in the terminal ileum. CAD findings P:6-P:15 are not shown. Below (right monitor), standard axial and optional coronal and sagittal 2D images (not shown). Optional 3D target views (not shown) of prone and supine datasets are used, first, to further evaluate potential polyp candidates or CAD findings on complex structures and, second, to perform a complete 2D review of the prone and the supine CTC datasets. The 2D axial prone and supine CT images show the polyp (crosshair) marked by CAD (marker 1) in both positions. All images courtesy of Dr. Thomas Mang.
CAD first-reader gallery evaluation shown in a screening patient with a 10-mm tubular adenoma in the rectum. Above (left monitor), all 15 CAD markers in a volume are presented to the reader, line by line, as a collection of CT image galleries. They are sorted top to bottom by decreasing level of confidence, as computed by the CAD algorithm both for the prone (shown) or for the supine (not shown) position. CAD finding P:1 (first row of images) displays the polyp (crosshair) in all six gallery images. Note that the typical imaging characteristics of a polyp are already recognizable within the gallery images. The remaining CAD findings P:2-P:5 (image rows 2-5) display false-positive detections in the stomach (P:2), on the residual fluid layer (P:3), in the stomach (P:4), and in the terminal ileum. CAD findings P:6-P:15 are not shown. Below (right monitor), standard axial and optional coronal and sagittal 2D images (not shown). Optional 3D target views (not shown) of prone and supine datasets are used, first, to further evaluate potential polyp candidates or CAD findings on complex structures and, second, to perform a complete 2D review of the prone and the supine CTC datasets. The 2D axial prone and supine CT images show the polyp (crosshair) marked by CAD (marker 1) in both positions. All images courtesy of Dr. Thomas Mang.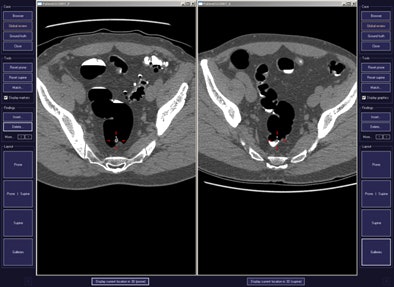
No limits on false positives
The CAD algorithm first looks for areas on the colon wall for further analysis, generating about 100 potential polyp locations per volume, Mang and his colleaues explained. It then calculates a set of image features, enters them into a classsifier that generates the polyp candidates, and orders them based on the computer probabilty of each mark corresponding to a true lesion, with a maximum of one to 15 candidates per image series. CAD findings are fixed and not adjustable by the reader. CTC testing cases were kept completely separate from training cases. The average calculation time was 8.7 minutes for 30 CAD marks and 0.67 minutes to generate the image galleries for each patient, the group reported.
A single experienced reader examined the data without knowing the reference-standard results, looking for adenomas 6 mm and larger, and limiting his search to CAD findings only. The study team calculated per-patient, per-polyp, and per-adenoma sensitivities for lesions 6 mm and larger. Each patient dataset was interpreted by initially using CAD image galleries followed by a fast 2D review to address unprompted colonic areas. Statistical tests included Fisher's exact and McNemar tests.
The CAD gallery offered a quick 2D and 3D visual overview of the CAD prompts to enable quick discrimination of true- from false-positive findings, the group stated. Each CAD finding was presented as an image series generated by the algorithm, based on the 3D coordinates of the CAD finding, and sorted in order of system confidence, the authors explained. Correlation of the prone and supine image galleries was facilitated by automated identificaiton of polyp candidates.
Good accuracy for new reading model
CTC and optical colonoscopy had matched findings for 131 polyps 6 mm or larger (92 adenomas, 39 nonadenomas) in 89 of 616 patients, while 527 patients had no polyps 6 mm or larger. The prevalance of polyps of any histology and adenomas 6 mm and larger was 14.4% in CTC and 10.4% in optical colonoscopy, and two patients without polyps had colorectal cancer, the authors reported.
Overall, CAD alone identified 86 of 89 (96.6 %) patients with polyps 6 mm or larger, 100% of patients with adenomas, and 88% of patients with nonadenomatous polyps (p = 0.02), Mang and colleagues reported.
Image-gallery-based first-reader CAD results
|
Using the image-gallery-based first-reader CAD approach, mean interpretation time was 3.1 minutes per patient. The positive predictive value for detecting patients with polyps 6 mm and larger was 76.5%, and the negative predictive value was 97.3%. The mean number of false-positive CAD detections was 29.8 per patient. In the CAD-guided read, 23 of 527 patients without polyps had false-positive detections, for a specificity of 95.6%. The maximum sensitivities for detecting adenomas 6 mm or larger were 89.1% per patient and 85.9 % per polyp. CAD found all 29 large adenomas and both cancers, all of which were correctly accepted by the reader.
Two small adenomas and three nonadenomas (including one large nonadenoma with flat morphology) were missed by both CAD and the reader. The most common reasons for dismissing true-positive CAD marks were low lesion conspicuity, usually due to flat lesion morphology, or CAD detection in only a single position, reducing confidence in the detection.
Overall, the CAD-first approach found 75 of 89 patients with polyps 6 mm and larger, with higher sensitivities for adenomas versus nonadenomas. The authors suggested that that this effect, also seen in previous studies, might be related to the lower conspicuity of nonadenomatous polyps.
Unlike CAD algorithms used in second- or concurrent-reader mode, the CAD algorithm used in this study doesn't minimze false-positive findings in favor of maximal sensitivity, but rather orders all CAD findings from top to bottom based on system confidence, the group stated. The increased number of findings was well addressed by the gallery display system, which allowed the radiologist to time-efficiently dismiss the majority of obvious CAD false positives and focus on the small number of potential polyp candidates, Mang and colleagues wrote.
"Using a CAD system in a first-reader workflow may be of clinical relevance to make CTC more time-efficient when evaluating large numbers of screening patients, especially in colorectal cancer screening programs," they wrote. "The results of this study differ from previous opinions on the limitations and aspects of liability associated with first reading, since they indicate that an accurate and time-efficient evaluation, based on galleries with expert reader performance, could be feasible in a low-prevalence population. However, further research is needed to translate these initial research results to patient care."