Ultrasound remains the gold standard for monitoring Poly Implant Prothese (PIP) breast implants, but new French data suggest that dedicated silicone sequences in MRI can help detect problems, particularly if asymptomatic patients wish to keep their implants.
The jury is still out, however, given that any suspicion related to pain, swelling, or other symptoms will usually result in systematic removal of implants rather than lengthy investigations.
Rather than a clear rupture, a "perspiring" implant, where gel slowly leaks through the porous membrane, is the most common problem that surgeons report, according to Dr. Corinne Balleyguier, head of breast imaging in the department of radiology at the Institut Gustave Roussy (IGR)in Villejuif, France.
"Perspiration can't be confirmed or assessed by ultrasound, but it accounts for 60% of problems related to defective implants," she commented. "Ultrasound may depict a slight fluid slick around the implant, but this might be normal breast fluid or perspiring gel."
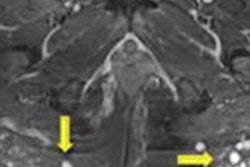
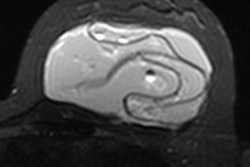
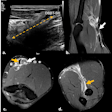
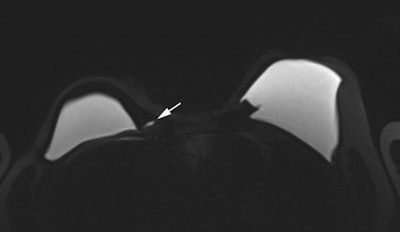 Top and bottom: Two breast MRI examinations acquired with a silicone dedicated sequence. Silicone gel appears bright, and fat and glandular tissue are dark. In cases of perspiration or rupture, an amount of bright fluid is seen outside of the implant (arrows), which facilitates diagnosis. Perspiration was not diagnosed on ultrasound in these two cases. Images courtesy of Dr. Corinne Balleyguier.
Top and bottom: Two breast MRI examinations acquired with a silicone dedicated sequence. Silicone gel appears bright, and fat and glandular tissue are dark. In cases of perspiration or rupture, an amount of bright fluid is seen outside of the implant (arrows), which facilitates diagnosis. Perspiration was not diagnosed on ultrasound in these two cases. Images courtesy of Dr. Corinne Balleyguier.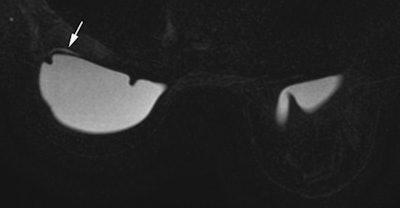
In research involving 270 implants removed at the IGR, MRI with dedicated silicone sequences was helpful in diagnosis of perspiration. In the 93 perspiring implants, 43 out of 49 ultrasound exams were normal, with six false positives for rupture. Sixteen MRI examinations were also performed on the 93 perspiring implants. Seven out of nine MRI exams showed perspiration because MRIs were acquired with silicone sequences; five out of seven MRI exams were normal, but these MRIs were acquired without silicone sequences.
Out of the 270 removed implants, there were 13 ruptures. Ultrasound was available for 12 of these, with six yielding normal imaging results and six abnormal. Only one MRI exam was available, and it was abnormal.
Because silicone was banned in France for many years, radiologists are not necessarily trained in imaging techniques for best depicting silicone implants, according to Dr. Anne Tardivon, breast imaging specialist in the department of radiology at the Curie Institute in Paris.
Where there are suspicious clinical symptoms but normal or equivocal ultrasound results, MRI may be used as a second line investigative tool. Access to MRI is problematic for French patients because there are proportionally fewer MRI units in France than in neighboring countries, such as Spain, Italy, and Belgium. A request for its inclusion in diagnosis depends largely on the discretion of the individual surgeon or clinician.
"French radiologists are better at performing ultrasound than they are MRI, and ultrasound will usually answer most questions. Given the high numbers of patients involved, using MRI highly selectively is a good compromise," she said, pointing to few diagnoses of implant abnormalities by MRI at the Curie Institute.
Tardivon has seen some of the hospital's 900 patients with PIP implants following surgery for breast cancer.
"We have simply added bi-yearly ultrasound and clinical examination to the yearly mammography. For patients with implants for cosmetic reasons, the follow-up may seem more burdensome. These women may be more likely to opt for surgery, to avoid the anxiety of 'waiting' for a rupture, or the risk of false negatives from ultrasound," noted Tardivon, who senses the French government would prefer all PIP implants to be removed over time, given its funding approval for explantation in all women who request it.
 Using MRI highly selectively is a good compromise, according to Dr. Anne Tardivon.
Using MRI highly selectively is a good compromise, according to Dr. Anne Tardivon.Because the gel is locally aggressive, extracapsular rupture yields early and clear clinical signs such as pain and swelling, which are easy to spot on ultrasound. Axillary lymph nodes must also be checked by ultrasound for siliconoma in case the implant is only slightly compromised, leading to slow leakage or a "perspiring" capsule, which does not give any clear clinical symptoms or radiological evidence in breast ultrasound, she explained.
After explantation in asymptomatic patients, some surgeons have reported that their gloves bore traces of silicone gel, showing the imaging to be a false negative for leaks, according to Tardivon.
Following December's meeting between the French Ministry of Health, National Cancer Institute (INCa) and the national agency for medical devices (AFSSAPS), the government announced its official strategy 12 months after the first alerts of detective implants were released to the medical community.
Asymptomatic women with PIP implants may choose to have them removed free-of-charge or undergo six-monthly clinical and ultrasound examinations, to be funded indefinitely by the French government. For those who opt for surgery, only breast cancer patients will benefit from free replacement implants.
The French authorities maintain that while PIP implants seem to present no extra risk of causing breast cancer or lymphoma, there is an increased risk of rupture, estimated at 5%, compared to the industry standard of 1-2%. Meanwhile, doctors are creating an imaging and histological database to gather as much information as possible for prospective studies on associated cancer risks as well as local complications in women who undergo surgery and in those that choose to keep their implants.
Both Tardivon and Balleyguier were members of the expert panel at December's multistakeholder meeting, which helped shape the recommendations of AFSSAPS and the Ministry of Health.
Nobody has yet estimated the cost to the French health budget of imaging follow-up or surgical removal of the implants in the 30,000 French women affected. Ultrasound costs nearly 42 Euros per examination, and indefinite bi-yearly imaging will prove costly, as will systematic surgical removal. An initial 5,000 surgical interventions have been -- or are planned to be -- performed, and the remainder may be carried out over the coming years.